WO2010147133A1 - 新規なイミダゾピリジン化合物 - Google Patents
新規なイミダゾピリジン化合物 Download PDFInfo
- Publication number
- WO2010147133A1 WO2010147133A1 PCT/JP2010/060172 JP2010060172W WO2010147133A1 WO 2010147133 A1 WO2010147133 A1 WO 2010147133A1 JP 2010060172 W JP2010060172 W JP 2010060172W WO 2010147133 A1 WO2010147133 A1 WO 2010147133A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- group
- indol
- imidazo
- naphthoic acid
- Prior art date
Links
- 0 CC1(*)C=CC=CC1c1c(Cc2c[n](*)c3c2c(*)c(*)cc3)[n](cccc2)c2n1 Chemical compound CC1(*)C=CC=CC1c1c(Cc2c[n](*)c3c2c(*)c(*)cc3)[n](cccc2)c2n1 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/14—Decongestants or antiallergics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
Definitions
- the present invention relates to an imidazopyridine compound, a salt thereof, an N-oxide thereof or a solvate thereof, or a prodrug thereof, and a medicine containing them as an active ingredient.
- an imidazopyridine compound, a salt thereof, an N-oxide thereof or a solvate thereof, or a prodrug thereof (hereinafter, these are generically named And a medicament containing them as active ingredients.
- Human chymase is a neutral serine protease isolated in 1990 and showing specificity for a chymotrypsin-like substrate, and has a molecular weight of about 30,000. Chymase is mainly present in granules in mast cells, and is secreted to the outside with degranulation of mast cells. Secreted chymase binds to an extracellular matrix such as heparan sulfate proteoglycan, thereby avoiding inhibition by in vivo inhibitors and exerts long-term enzyme activity in the heart, blood vessels, skin, and other tissues. Are known.
- ACE angiotensin converting enzyme
- chymase works as follows. Promotion of degranulation of mast cells, conversion of interleukin-1 ⁇ and interleukin-18 from pro form to active form, activation of MMPs such as matrix metalloproteinase (hereinafter abbreviated as MMP) -2 and MMP-9 Activation of transforming growth factor ⁇ (TGF- ⁇ ), release of stem cell factor (stem cell factor, SCF) expressed on the cell membrane from the cell membrane, endothelin-1 consisting of 31 amino acid residues from big endothelin-1 (1 -31).
- MMPs matrix metalloproteinase
- TGF- ⁇ transforming growth factor ⁇
- SCF stem cell factor
- Non-Patent Documents 1, 2, 3, and 4 In addition to being deeply involved in these various biological responses, chymase is involved in many pathologies such as atopic dermatitis, ulcerative colitis, heart failure, pulmonary fibrosis, and post-surgical organ adhesions. It has also been suggested by animal experiments (Non-Patent Documents 1, 2, 3, and 4).
- the chymase inhibitor may become a new type of cardiovascular disease therapeutic agent, anti-inflammatory agent and anti-allergic agent. ing.
- Patent Document 1 includes general formula (Ia)
- R 1 a and R 2 a are each independently a hydrogen atom, halogen, trihaloalkyl, C 1-4 alkyl, etc .
- a a is halogen, C 1-6 linear or branched alkyl, C 1-6 linear or branched alkoxy, C 1-6 linear or branched alkylthio, C 1-6 straight One or more —O—, —S—, —SO—, —SO 2 — optionally substituted with 1 to 3 substituents selected from linear or branched alkylsulfonyl, phenyl and oxo
- E a is —COORe a or the like
- Each of Re a is independently a hydrogen atom, C 1-4 alkyl or aryl
- G a is C 1-6 linear or branched alkyl optionally incorporated by one or more —O—,
- Patent Document 2 includes general formula (Ib).
- R 1b and R 2b are simultaneously or independently hydrogen atom, halogen atom, trihalomethyl group, alkyl group having 1 to 4 carbon atoms, etc .;
- Ab is a substituted or unsubstituted arylene group having 6 to 11 carbon atoms, or a substituted or unsubstituted oxygen, nitrogen and sulfur atom which may contain one or more atoms on the ring.
- a heteroarylene group; E b is —COOR 3b or the like; R 3b represents a hydrogen atom or a linear or branched alkyl group having 1 to 6 carbon atoms; G b represents a substituted or unsubstituted linear or branched alkylene group having 1 to 6 carbon atoms; M b is a single bond or S (O) m b ; m b is an integer from 0 to 2; Jb is a substituted or unsubstituted aryl group having 6 to 11 carbon atoms, or a heterocycle having 4 to 10 carbon atoms which may contain one or more substituted or unsubstituted oxygen, nitrogen and sulfur atoms on the ring.
- Patent Document 3 discloses general formula (Ic).
- R 1c and R 2c are simultaneously or independently hydrogen atom, halogen atom, trihalomethyl group, alkyl group having 1 to 4 carbon atoms, etc .; Ac has one or more heteroatoms selected from the group consisting of a substituted or unsubstituted arylene group having 6 to 11 carbon atoms, or a substituted or unsubstituted oxygen atom, nitrogen atom and sulfur atom on the ring.
- a heteroarylene group having 4 to 10 carbon atoms; E c is —COOR 3c and the like; R 3c represents a hydrogen atom or a linear or branched alkyl group having 1 to 6 carbon atoms; G c represents a substituted or unsubstituted linear or branched alkylene group having 1 to 6 carbon atoms, and the like; M c is a single bond or -S (O) m c -; m c is an integer from 0 to 2; J c, if M c is a single bond, a substituted or unsubstituted aryl group having a carbon number of 6-11, or a substituted or unsubstituted oxygen atoms, one or more selected from the group consisting of nitrogen atom and a sulfur atom A heteroaryl group having 4 to 10 carbon atoms having a heteroatom on the ring; X c is —CH ⁇ or a nitrogen atom; Y c is —CB
- the chymase inhibitors disclosed so far have not been satisfactory in terms of enzyme inhibitory activity, safety, and metabolic stability.
- a chymase inhibitor effective for chymase-mediated diseases such as skin diseases, circulatory system diseases, digestive system diseases, respiratory system diseases, liver diseases, eye diseases, etc., and has extremely potent chymase inhibitory activity Therefore, a drug having a large gap (ratio) between chymase inhibitory activity and toxicity, that is, higher safety and high metabolic stability is desired.
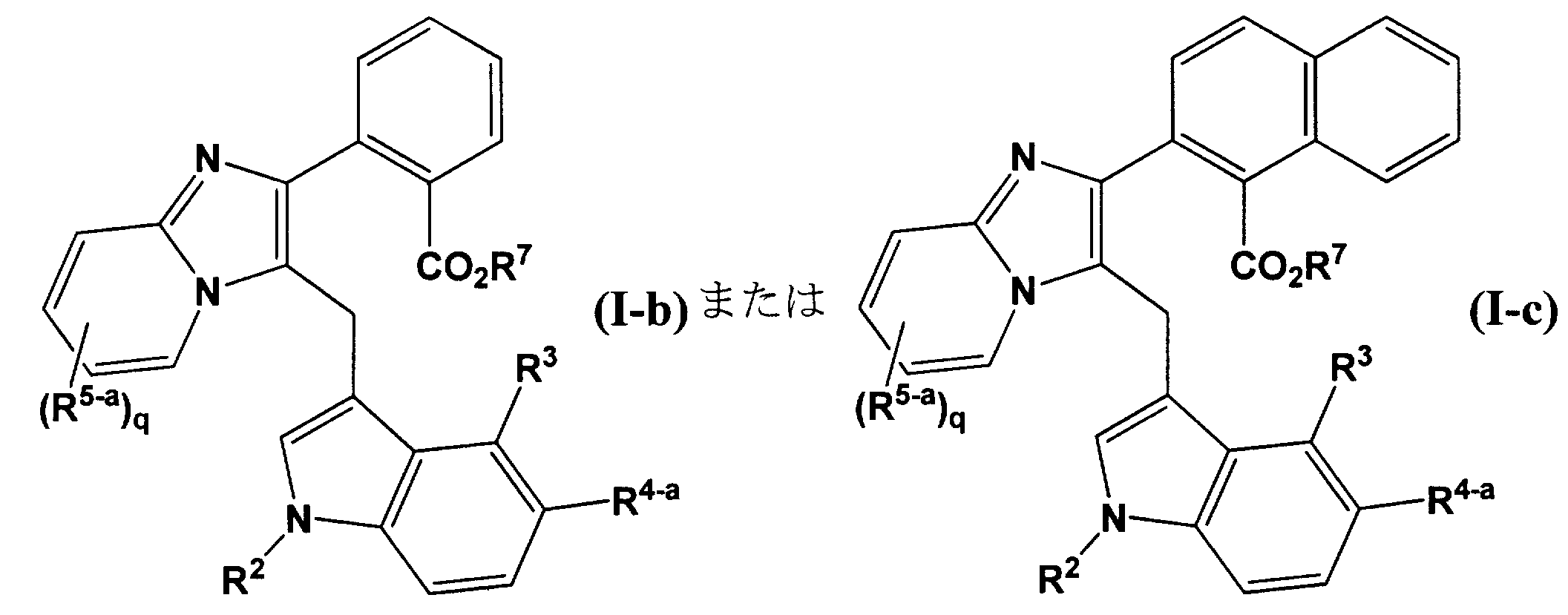
- R 1 represents a hydrogen atom, a methyl group, a trifluoromethyl group, a difluoromethyl group, a fluorine atom, or a chlorine atom
- R 2 is a hydrogen atom
- an alkyl group of one to six are C1 may be -10 substituted with R 11, 1-6 alkenyl group which may be C2-10 substituted by R 11 or, Represents a C2-10 alkynyl group optionally substituted by 1-6 R 11
- R 11 is (I) a halogen atom
- R 8 is (1) a hydrogen atom, (2) a C1-3 alkyl group, (3) a C1-3 haloalkyl group, (4) C5-6 carbocyclic group, (5) a 5- to 6-membered heterocyclic group containing 1 to 2 nitrogen atoms
- NR 9 R 10 (wherein R 9 and R 10 each independently represents a hydrogen atom, a C1-3 alkyl group, or a phenyl group), (Iv) a C5-6 carbocyclic group optionally substituted with at least one group selected from a halogen atom, an oxo group, and a methyl group, or (v) selected from a halogen atom, an oxo group, and a methyl group Containing 1 to 4 nitrogen atoms, 1 to 2 oxygen atoms and / or 1 to 2 sulfur atoms that may be oxidized, which may be substituted with at least one group selected from 3 to Represents a 10-membered heterocyclic group, R 3 represents a hydrogen atom, a methyl group, or a fluorine atom, R 4 represents a hydrogen atom, a C1-2 alkyl group, a C1-2 haloalkyl group, or a halogen atom, R 5 represents
- R 4 , R 5 , and R 6 may be independently the same or different.
- R 4-a is a hydrogen atom, a methyl group, a fluorine atom or a chlorine atom
- R 5-a represents a hydrogen atom, a methyl group or a fluorine atom
- the [ 1] represents the same meaning as the symbol described above, and the compound according to [1] above, [3]
- R 2 is a hydrogen atom
- an alkyl group of one to six are C1 may be -10 substituted with R 12, 1-6 alkenyl which may be optionally C2 ⁇ 10 substituted by R 12 Or a C2-10 alkynyl group optionally substituted by 1 to 6 R 12
- R 12 may be substituted with a halogen atom, a hydroxyl group, a dimethylamino group, a pyrrolidinyl group, an N-methylpyrrolidinyl group, a piperidinyl group, a phenoxy group which may be substituted with a halogen atom, or a halogen atom.
- R 2 is an alkyl group of one to six C1-6 substituted with R
- a method for preventing and / or treating chymase-mediated diseases [14] A compound represented by the general formula (I), a salt thereof, an N-oxide thereof, or a solvate thereof, or a solvate thereof for preventing and / or treating a chymase-mediated disease Prodrugs, [15] A compound represented by the general formula (I) according to the above [1], a salt thereof, an N-oxide thereof or a solvate thereof for the manufacture of a prophylactic and / or therapeutic agent for chymase-mediated diseases Or the use of their prodrugs.
- the compound of the present invention exhibits an extremely strong chymase inhibitory activity that has never been seen before. Furthermore, the compound of the present invention has a very large divergence (ratio) between chymase inhibitory activity and hepatotoxicity, so that it is a drug that is greatly reduced with respect to the side effect risk of hepatotoxicity. In addition, the compound of the present invention is excellent in metabolic stability. Therefore, the compound of the present invention is a drug having all the profiles such as sufficient chymase inhibitory activity, higher safety and high metabolic stability.
- the “6- to 10-membered monocyclic aromatic heterocycle” in the “6- to 10-membered monocyclic or bicyclic aromatic heterocycle” includes, for example, pyridine, pyridazine, pyrimidine, It is a pyrazine ring.
- the “6- to 10-membered bicyclic aromatic heterocycle” in the “6- to 10-membered monocyclic or bicyclic aromatic heterocycle” includes, for example, quinoline, isoquinoline, cinnoline, Quinazoline and quinoxaline rings.
- the “C1-10 alkyl group optionally substituted with R 11 ” represented by R 2 means “straight chain or branched C1-10”. Is an alkyl group.
- Examples of the “linear or branched C1-10 alkyl group” include, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, 1 -Methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-ethylpropyl, 2-ethylpropyl, 2,2-dimethylpropyl, n-hexyl, 1-methylpentyl 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-d
- the “C2-10 alkenyl group” in the “C2-10 alkenyl group optionally substituted by R 11 ” represented by R 2 means “straight chain or branched C2-10”. Is an alkenyl group.
- Examples of the “linear or branched C2-10 alkenyl group” include, for example, vinyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 3-butenyl and 1-methyl.
- -1-propenyl 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1,2-dimethylvinyl, 1-ethylvinyl, 1- Pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl- 2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1,2-dimethyl-1-propenyl, 1- Ethyl-1-propenyl, 1-eth -2-propenyl, 2-ethyl-2-propenyl, 1,1-dimethyl-2-propenyl, 1-propylvinyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-
- the “C2-10 alkynyl group” in the “C2-10 alkynyl group optionally substituted with R 11 ” represented by R 2 means “straight chain or branched C2-10”. Is an alkynyl group.
- “Straight or branched C2-10 alkynyl group” means, for example, ethynyl, 1-propynyl, 2-propynyl, 1-methyl-2-propynyl, 1-butynyl, 2-butynyl, 3-butynyl 1-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 3-methyl-1-butynyl, 1-methyl-2-butynyl, 2-methyl-3-butynyl, 1-methyl-3-butynyl, 1 -Hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 4-methyl-1-pentynyl, 3-methyl-1-pentynyl, 4-methyl-2-pentynyl, 1-methyl-2-pentynyl 2-methyl-3-pentynyl, 1-methyl-3-pentynyl,
- alkyl C1 to 10 in the “alkyl group having a C1 may be -10 substituted with R 12" represented by R 2, optionally substituted with "R 11 represented by R 2 “C1-10 alkyl group” in “optionally C1-10 alkyl group”.
- alkenyl group C2 ⁇ 10 in the "alkenyl group which may be C2 ⁇ 10 substituted with R 12" represented by R 2, optionally substituted with "R 11 represented by R 2 “C2-10 alkenyl group” in “optionally C2-10 alkenyl group”.
- alkynyl group C2 ⁇ 10 in the "alkynyl group which may be C2 ⁇ 10 substituted with R 12" represented by R 2, optionally substituted with "R 11 represented by R 2 And “C2-10 alkynyl group” in “optionally C2-10 alkynyl group”.
- alkyl group of C1 ⁇ 6 in the “alkyl group of C1 ⁇ 6 that is substituted with R 12" represented by R 2, "linear or branched alkyl of C1 ⁇ 6 Group.
- Examples of the “linear or branched C1-6 alkyl group” include, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, 1 -Methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-ethylpropyl, 2-ethylpropyl, 2,2-dimethylpropyl, n-hexyl, 1-methylpentyl 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1,1-dimethylbutyl, 1,2-
- the “C2-6 alkenyl group” in the “C2-6 alkenyl group optionally substituted by R 12 ” represented by R 2 means “straight chain or branched C2-6”. Is an alkenyl group.
- Examples of the “linear or branched C2-6 alkenyl group” include, for example, vinyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 3-butenyl, and 1-methyl.
- -1-propenyl 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1,2-dimethylvinyl, 1-ethylvinyl, 1- Pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl- 2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1,2-dimethyl-1-propenyl, 1- Ethyl-1-propenyl, 1-ethyl 2-propenyl, 2-ethyl-2-propenyl, 1,1-dimethyl-2-propenyl, 1-propylvinyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5
- the “C2-6 alkynyl group” in the “C2-6 alkynyl group optionally substituted by R 12 ” represented by R 2 means “straight chain or branched C2-6”. Is an alkynyl group.
- “Straight or branched C2-6 alkynyl group” means, for example, ethynyl, 1-propynyl, 2-propynyl, 1-methyl-2-propynyl, 1-butynyl, 2-butynyl, 3-butynyl 1-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 3-methyl-1-butynyl, 1-methyl-2-butynyl, 2-methyl-3-butynyl, 1-methyl-3-butynyl, 1 -Hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 4-methyl-1-pentynyl, 3-methyl-1-pentynyl, 4-methyl-2-pentynyl, 1-methyl-2-pentynyl 2-methyl-3-pentynyl, 1-methyl-3-pentynyl,
- the “C1-2 alkyl group” represented by R 4 is a methyl or ethyl group.
- the “C1-3 alkyl group” represented by R 5 , R 6 , R 8 , R 9 , and R 10 is a methyl, ethyl, n-propyl, or isopropyl group.
- the “C1-4 alkyl group” represented by R 7 is a methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, or isobutyl group.
- the “C1-3 haloalkyl group” represented by R 5 , R 6 and R 8 is, for example, monofluoromethyl, difluoromethyl, trifluoromethyl, 2-fluoroethyl, 1-fluoroethyl, 2,2-difluoroethyl, 1,2-difluoroethyl, 1,1-difluoroethyl, 2,2,2-trifluoroethyl, 1,2,2-trifluoroethyl, 1,1,2-trifluoroethyl 1,2,2,2-tetrafluoroethyl, 1,1,2,2-tetrafluoroethyl, pentafluoroethyl, 1,2-dibromo-1,2,2-trifluoroethyl, 1-chloro-1 , 2,2,2-tetrafluoroethyl, 3-fluoropropyl, 3-chloropropyl, 2-fluoropropyl, 2-chloropropyl, 2-chloro
- the “C1-2 haloalkyl group” represented by R 4 is, for example, monofluoromethyl, difluoromethyl, trifluoromethyl, 2-fluoroethyl, 1-fluoroethyl, 2,2-difluoroethyl, 1,2-difluoroethyl, 1,1-difluoroethyl, 2,2,2-trifluoroethyl, 1,2,2-trifluoroethyl, 1,1,2-trifluoroethyl, 1,2,2, 2-tetrafluoroethyl, 1,1,2,2-tetrafluoroethyl, pentafluoroethyl, 1,2-dibromo-1,2,2-trifluoroethyl, and 1-chloro-1,2,2,2 A tetrafluoroethyl group.
- the “C1-3 alkoxy group” represented by R 8 is, for example, a methoxy, ethoxy, n-propoxy, or isopropoxy group.
- the “C5-6 carbocyclic group” represented by R 8 is, for example, a cyclopentyl, cyclopentenyl, cyclopentadienyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, and phenyl group.
- the “group” include “C5-6 carbocyclic group” represented by R 8 .
- 3- to 10-membered heterocyclic group containing means, for example, aziridinyl, oxiranyl, thiranyl, azetidinyl, oxetanyl, thietanyl, azepanyl, oxepanyl, thiepanyl, 1,4-diazepanyl, 1,4-oxazepanyl, azacyclo Octanyl, 1,5-diazacyclooctanyl, pyrrolidyl, tetrahydrofuranyl, thiolanyl, sulfolanyl, piperidinyl, tetrahydropyra Nyl, tetrahydrothiopyranyl, 1,1-dioxohexahydrothiopyranyl, hexahydropyrimidinyl, piperazinyl, morpholinyl, thiomorpholinyl, 1,1-dioxothiomorpholin
- R 12 is a halogen atom, a hydroxyl group, a dimethylamino group, a pyrrolidinyl group, an N-methylpyrrolidinyl group, a piperidinyl group, a phenoxy group optionally substituted with a halogen atom, or a halogen atom.
- phenyl group tetrahydropyranyl group, 2,3-dihydrobenzofuranyl group, thiophenyl group, diisopropylamino group, methyl (phenyl) amino group, piperazinyl group, 2-oxo-1-pyrrolidinyl group Morpholinyl, 1,1-dioxothiomorpholinyl, imidazolyl, pyridyl, 2-methoxyethyleneoxy, dimethylaminoethyleneoxy, diethylaminoethyleneoxy, 1,2,3,4-tetrahydroiso Quinolinyl group, azepanyl group, pyrazolyl group, 2-oxoimidazolidini Group, diethylamino group, methoxy group, and N-methylpyrazolyl group.
- halogen atom refers to fluorine, chlorine, bromine, and iodine atoms.
- R 1 is preferably a hydrogen atom.
- a hydrogen atom, an alkyl group of one to six are C1 may be -10 substituted with R 12, 1-6 may be substituted with R 12 C2 ⁇ 10 alkenyl groups, or a C2-10 alkynyl group optionally substituted by 1 to 6 R 12 , more preferably a hydrogen atom, a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an alkyl group of one to six C1 ⁇ 6 that is substituted with R 12, substituted with 1-6 alkenyl group may C2 ⁇ 6 to be substituted by R 12 or 1-6 R 12, An optionally substituted C2-6 alkynyl group, more preferably a hydrogen atom, a methyl group, an ethyl group, or a 2- (4-morpholinyl) ethyl group.
- R 3 is preferably a hydrogen atom or a methyl group.
- R 3 is more preferably a methyl group.
- R 4 is preferably a hydrogen atom, a methyl group, a fluorine atom, or a chlorine atom.
- R 4 is more preferably a fluorine atom.
- R 5 is preferably a hydrogen atom, a methyl group, or a fluorine atom.
- R 5 and more preferably include a hydrogen atom or a fluorine atom.
- R 6 is preferably a hydrogen atom, a methyl group, or a fluorine atom.
- R 6 is more preferably a hydrogen atom.
- R 7 is preferably a hydrogen atom or a methyl group.
- R 7 is more preferably a hydrogen atom.
- R 8 is a hydrogen atom, a C1-3 alkyl group, a C5-6 carbocyclic group, a C1-3 alkyl group substituted with a C1-3 alkoxy group, or a NR 9 R 10 substituent. Preferred are C1-3 alkyl groups. R 8 is more preferably a hydrogen atom, a methyl group, a phenyl group, a methoxyethyl group, or an ethyl group substituted with NR 9 R 10 .
- R 9 and R 10 are preferably a methyl group, an ethyl group, an isopropyl group, or a phenyl group.
- R 11 represents a fluorine atom, a hydroxyl group, a dimethylamino group, a pyrrolidinyl group, an N-methylpyrrolidinyl group, a piperidinyl group, a phenoxy group, a phenyl group, a phenyl group substituted with a fluorine atom, a tetrahydropyrani group.
- R 12 represents a fluorine atom, a hydroxyl group, a dimethylamino group, a pyrrolidinyl group, an N-methylpyrrolidinyl group, a piperidinyl group, a phenoxy group, a phenyl group, a phenyl group substituted with a fluorine atom, a tetrahydropyrani group.
- R 4-a is preferably a hydrogen atom, a methyl group, a fluorine atom, or a chlorine atom.
- R 4-a is more preferably a fluorine atom.
- R 5-a is preferably a hydrogen atom, a methyl group, or a fluorine atom.
- R 5-a is more preferably a hydrogen atom or a fluorine atom.
- the ring represented by is preferably a benzene, naphthalene, pyridine, pyrimidine, pyridazine, pyrazine, quinoline, or isoquinoline ring, and more preferably a benzene or naphthalene ring.
- the compound represented by the general formula (I) is preferably
- alkyl groups include straight chain and branched chain.
- isomers R, S form, ⁇ , ⁇ configuration, enantiomers, diastereomers
- optically active substances having optical activity D, L, d, 1 form
- chromatographic separation due to the presence of asymmetric carbon etc.
- a polar form high polar form, low polar form
- an equilibrium compound a rotational isomer, a mixture of these in any proportion, and a racemic mixture are all included in the present invention.
- the salt is preferably a pharmaceutically acceptable salt.
- the salt is preferably water-soluble.
- the salt examples include alkali metal salts, alkaline earth metal salts, ammonium salts, amine salts, and acid addition salts.
- alkali metal salt examples include potassium and sodium.
- alkaline earth metal salt examples include calcium and magnesium.
- ammonium salts examples include tetramethylammonium.
- amine salts include triethylamine, methylamine, dimethylamine, cyclopentylamine, benzylamine, phenethylamine, piperidine, monoethanolamine, diethanolamine, tris (hydroxymethyl) aminomethane, lysine, arginine, and N-methyl-D- Examples include glucamine.
- acid addition salts include inorganic acid salts such as hydrochloride, hydrobromide, hydroiodide, sulfate, phosphate, nitrate, or acetate, lactate, tartrate, benzoic acid.
- Organic acid salts such as salt, citrate, methanesulfonate, ethanesulfonate, trifluoroacetate, benzenesulfonate, toluenesulfonate, isethionate, glucuronate, or gluconate Can be mentioned.
- the compound of the present invention can be converted into an N-oxide form by an arbitrary method.
- the N-oxide form represents an oxidized form of the nitrogen atom of the compound represented by the general formula (I).
- the compound represented by the general formula (I) and a salt thereof can be converted into a solvate.
- the solvate is preferably non-toxic and water-soluble.
- Suitable solvates include, for example, solvates such as water or alcohol solvents (for example, ethanol).
- the prodrug of the compound represented by the general formula (I) refers to a compound that is converted into a compound represented by the general formula (I) by a reaction with an enzyme, gastric acid or the like in a living body.
- the prodrug of the compound represented by the general formula (I) includes a compound in which the carboxy group represented by the general formula (I) is esterified or amidated (for example, the carboxy group of the compound represented by the general formula (I)).
- the group is ethyl esterified, isopropyl esterified, phenyl esterified, carboxymethyl esterified, dimethylaminomethyl esterified, pivaloyloxymethyl esterified, ethoxycarbonyloxyethyl esterified, phthalidyl esterified, (5-methyl- 2-oxo-1,3-dioxolen-4-yl) methyl esterification, cyclohexyloxycarbonylethyl esterification, methylamidated compounds, etc.); the carboxyl group represented by the general formula (I) is replaced with a hydroxymethyl group And the like.
- the prodrug of the compound represented by the general formula (I) may be either a hydrate or a non-hydrate.
- the compound represented by the general formula (I) is an isotope (for example, 2 H, 3 H, 11 C, 13 C, 14 C, 13 N, 15 N, 15 O, 17 O, 18 O, 35 S, 18 F, 36 Cl, 123 I, 125 I, etc.).
- the compound of the present invention represented by the general formula (I) can be produced according to a known method, for example, a method shown below, a method analogous thereto or a method shown in Examples.
- the raw material compound may be used as a salt.
- the reactive functional group such as a carboxyl group
- the reactive functional group can be protected with an appropriate protective group as necessary, and the protective group can be removed after the reaction is completed.
- a raw material compound is used as a salt, what was described as a pharmaceutically acceptable salt of general formula (I) is used as this salt.
- the carbon-carbon bond forming reaction between the compound represented by the general formula (II) and the compound represented by the general formula (III) can be performed by, for example, acid (organic acid (acetic acid, trifluoroacetic acid, methanesulfonic acid, p-toluenesulfone).
- acid organic acid (acetic acid, trifluoroacetic acid, methanesulfonic acid, p-toluenesulfone).
- Examples of the protecting group for the carboxyl group include methyl group, ethyl group, allyl group, tert-butyl group, trichloroethyl group, benzyl (Bn) group, phenacyl group, p-methoxybenzyl group, trityl group, 2-chlorotrityl group.
- Examples thereof include a solid phase carrier to which a group or a structure thereof is bound.
- the protective group for the carboxyl group is not particularly limited as long as it is a group that can be easily and selectively eliminated other than the above.
- those described in T. W. Greene, Protective Groups in Organic Synthesis, Wiley, New York, 1999 are used.
- the deprotection reaction of the protecting group of the carboxyl group is well known, for example, (1) Deprotection reaction by alkali hydrolysis, (2) Deprotection reaction under acidic conditions, (3) Deprotection reaction by hydrogenolysis, (4) desilylation reaction, (5) Deprotection reaction using metal, (6) Deprotection reaction using a metal complex and the like.
- the deprotection reaction by alkali hydrolysis is carried out, for example, in an organic solvent (methanol, tetrahydrofuran, dioxane, etc.), alkali metal hydroxide (sodium hydroxide, potassium hydroxide, lithium hydroxide, etc.), alkaline earth
- the reaction is carried out at a temperature of about 0 to 150 ° C. using a metal hydroxide (barium hydroxide, calcium hydroxide, etc.) or carbonate (sodium carbonate, potassium carbonate, etc.), an aqueous solution thereof or a mixture thereof.
- the deprotection reaction under acidic conditions can be performed by, for example, using an organic acid (acetic acid, trifluoroacetic acid, methanesulfonic acid, p-toluenesulfonic acid) in an organic solvent (dichloromethane, chloroform, dioxane, ethyl acetate, anisole, etc.). Etc.), or in an inorganic acid (hydrochloric acid, sulfuric acid, etc.) or a mixture thereof (hydrogen bromide / acetic acid, etc.) in the presence or absence of 2,2,2-trifluoroethanol, a temperature of about 0-100 ° C. Is done.
- Deprotection reaction by hydrogenolysis includes, for example, solvent (ether (tetrahydrofuran, dioxane, dimethoxyethane, diethyl ether, etc.), alcohol (methanol, ethanol, etc.), benzene (benzene, toluene, etc.), ketone System (acetone, methyl ethyl ketone, etc.), nitrile (acetonitrile, etc.), amide system (dimethylformamide, etc.), water, ethyl acetate, acetic acid or a mixed solvent of two or more thereof, catalyst (palladium-carbon, palladium black, In the presence of palladium hydroxide-carbon, platinum oxide, Raney nickel, etc.), in a hydrogen atmosphere under normal pressure or under pressure, or in the presence of ammonium formate, at a temperature of about 0 to 200 ° C.
- solvent ether (tetrahydrofuran, dioxane, dimethoxyethane
- the desilylation reaction is performed at a temperature of about 0 to 40 ° C. using tetrabutylammonium fluoride in an organic solvent miscible with water (tetrahydrofuran, acetonitrile, etc.), for example.
- the deprotection reaction using a metal is performed, for example, in an acidic solvent (acetic acid, a buffer solution of pH 4.2 to 7.2 or a mixed solution thereof with an organic solvent such as tetrahydrofuran) in the presence of powdered zinc, If necessary, it is performed at a temperature of about 0 to 40 ° C. while applying ultrasonic waves.
- an acidic solvent acetic acid, a buffer solution of pH 4.2 to 7.2 or a mixed solution thereof with an organic solvent such as tetrahydrofuran
- the deprotection reaction using a metal complex is carried out, for example, in an organic solvent (dichloromethane, dimethylformamide, tetrahydrofuran, ethyl acetate, acetonitrile, dioxane, ethanol, etc.), water or a mixed solvent thereof with a trap reagent (tributyltin hydride, Triethylsilane, dimedone, morpholine, diethylamine, pyrrolidine, etc.), organic acids (acetic acid, formic acid, 2-ethylhexanoic acid, etc.) and / or organic acid salts (sodium 2-ethylhexanoate, potassium 2-ethylhexanoate, etc.) In the presence, in the presence or absence of phosphine reagents (triphenylphosphine, etc.), metal complexes (tetrakistriphenylphosphine palladium (0), bis (tripheny
- the deprotection reaction can be performed by the method described in, for example, T. W. Greene, Protective Group, Organic, Synthesis, Wiley, New York, 1999.
- the compound represented by the general formula (II) can be produced by the method represented by the reaction process formula 1.
- X represents a halogen atom or a sulfonate (for example, mesylate, tosylate, trifluoromethanesulfonate, etc.), and other symbols have the same meanings as those described in [1] above.
- reaction scheme 1 The Heck reaction, acid hydrolysis reaction, ⁇ -bromination reaction, cyclization reaction, formylation reaction, and reduction reaction represented by reaction scheme 1 are the conditions of the examples described in the present specification, or It can be performed under known conditions.
- the compound represented by the general formula (III) is known, or known methods such as “ComprehensivehenOrganic Transformations: A Guide to Functional Group Preparations 2nd Edition (Richard C. Larock, John Wiley & Sons Inc, 1999) Can be easily produced by using the method described in the above.
- reaction involving heating can be performed using a water bath, an oil bath, a sand bath, or a microwave, as will be apparent to those skilled in the art.
- a solid-phase-supported reagent supported on a polymer for example, polystyrene, polyacrylamide, polypropylene, polyethylene glycol, etc.
- a polymer for example, polystyrene, polyacrylamide, polypropylene, polyethylene glycol, etc.
- the reaction product is obtained by a conventional purification means such as distillation under normal pressure or reduced pressure, high performance liquid chromatography using silica gel or magnesium silicate, thin layer chromatography, ion exchange resin, It can be purified by scavenger resin, column chromatography, washing, recrystallization or the like. Purification may be performed for each reaction or after completion of several reactions.
- a conventional purification means such as distillation under normal pressure or reduced pressure, high performance liquid chromatography using silica gel or magnesium silicate, thin layer chromatography, ion exchange resin, It can be purified by scavenger resin, column chromatography, washing, recrystallization or the like. Purification may be performed for each reaction or after completion of several reactions.
- the compound of the present invention Since the compound of the present invention exhibits chymase inhibitory activity, it is suitable for chymase inhibition mediated diseases such as skin diseases, cardiovascular diseases, gastrointestinal diseases, respiratory diseases, kidney diseases, eye diseases, liver diseases, and surgery. It is useful as a preventive and / or therapeutic agent for organ adhesion caused by the disease.
- examples of skin diseases include atopic dermatitis, allergic contact dermatitis, scleroderma, systemic scleroderma, keloid, hypertrophic scar, urticaria, psoriasis and the like.
- cardiovascular diseases include hypertension, pulmonary hypertension, cardiomyopathy, myocarditis, restenosis after percutaneous coronary angioplasty and / or thrombolysis, stenosis after placement of artificial blood vessels and / or transplanted blood vessels ,
- Heart failure eg, congestive heart failure, acute heart failure, chronic heart failure
- arrhythmia eg, angina (eg, unstable angina)
- myocardial infarction eg, acute myocardial infarction
- coronary artery examples include diseases, arteriosclerosis (for example, obstructive arteriosclerosis), peripheral circulatory disorder, cerebrovascular disease, vasculitis, aneurysm, vascular disorder of arteriovenous fistula during hemodialysis, and the like.
- digestive system diseases include peptic ulcer (eg, gastric ulcer, duodenal ulcer, small intestine ulcer, non-steroidal anti-inflammatory analgesic peptic ulcer), inflammatory bowel disease (eg, ulcerative colitis, Crohn's disease) Etc.).
- respiratory diseases include bronchial asthma, chronic obstructive pulmonary disease, chronic bronchitis, emphysema, idiopathic interstitial pneumonia, pulmonary fibrosis (for example, idiopathic pulmonary fibrosis), and the like.
- the renal disease include diabetic nephropathy or non-diabetic nephropathy, IgA nephropathy, glomerulonephritis and the like.
- Examples of the eye disease include myopia, dry eye, corneal epithelial disorder, blepharitis, allergic conjunctivitis and the like.
- liver diseases include cirrhosis, liver fibrosis, nonalcoholic fatty liver disease (eg, nonalcoholic steatohepatitis), alcoholic steatohepatitis, and the like.
- organ adhesion resulting from surgery include abdominal organ adhesion after abdominal surgery, adhesion after glaucoma surgery (eg trabeculectomy), adhesion after craniotomy (eg external decompression, etc.), Examples include adhesion after cardiac surgery.
- the compound of the present invention not only has an unprecedented strong chymase inhibitory activity, but also has a very low risk of hepatotoxic side effects and is excellent in metabolic stability, and thus becomes a clinically useful drug.
- chymase-mediated diseases are chronic diseases, especially long-term drugs to treat atopic dermatitis, ulcerative colitis, peptic ulcer, nonalcoholic fatty liver disease, and pulmonary fibrosis Since it will be taken, it is desirable to find a drug with a reduced burden on the liver. That is, a compound with a low liver toxicity risk is desired.
- the hepatotoxicity risk can be evaluated by, for example, mitochondrial function, reactive metabolite, human liver cancer cell line (abbreviated as HepG2) and the like.
- the ratio between the main activity (IC 50 ) and, for example, HepG2 cytotoxicity (IC 50 ) (IC 50 (HepG2 cytotoxicity) / IC 50 (main activity of pharmaceuticals)) is about 0.06 to 2000. Therefore, a compound having a ratio value of about 2000 is desired as a compound having a low liver toxicity risk.
- the ratio between the cytotoxicity (IC 50 ) of HepG2 and the chymase inhibitory activity (IC 50 ) (IC 50 (cytotoxicity of HepG2)) / IC 50 (chymase inhibitory activity)) is preferably 2000 times or more, more preferably 3500 times or more.
- chymase-mediated diseases especially atopic dermatitis, ulcerative colitis, peptic ulcer, non-alcoholic fatty liver disease, and pulmonary fibrosis
- IC 90 an index showing the maximum action.
- HepG2 cytotoxicity should be evaluated by IC 10 so as to find a safety risk in the liver more sensitively in order to find a drug with a reduced burden on the liver.
- the compound of the present invention is useful as a preventive and / or therapeutic agent for hyperuricemia.
- the compound of the present invention 1) complementation and / or enhancement of the prophylactic and / or therapeutic effect of the compound, 2) Improving the kinetics / absorption of the compound, reducing the dose, and / or 3) reducing the side effects of the compound may be combined with other drugs and administered as a concomitant drug.
- the concomitant drug of the compound of the present invention and another drug may be administered in the form of a combination drug containing both components in one preparation, or may be administered in separate preparations.
- simultaneous administration and administration by time difference are included.
- administration with a time difference may be such that the compound of the present invention is administered first and the other drug may be administered later, or the other drug may be administered first and the compound of the present invention may be administered later.
- Each administration method may be the same or different.
- the disease that exerts a preventive and / or therapeutic effect by the above concomitant agent is not particularly limited as long as it is a disease that complements and / or enhances the preventive and / or therapeutic effect of the compound of the present invention.
- Examples of other drugs for complementation and / or enhancement of the preventive and / or therapeutic effect of the compound of the present invention on atopic dermatitis include, for example, non-steroidal anti-inflammatory drugs, steroid drugs, immunosuppressive drugs, prostaglandins , Antiallergic agents, mediator release inhibitors, leukotriene receptor antagonists, antihistamines, opioid agonists (eg, ⁇ -opioid agonists), phosphodiesterase inhibitors, forskolin preparations, nitric oxide synthase inhibitors, And cannabinoid-2 receptor stimulants, decoy preparations such as NF- ⁇ B, and chymase inhibitors.
- non-steroidal anti-inflammatory drugs include, for example, steroid drugs, immunosuppressive drugs, prostaglandins , Antiallergic agents, mediator release inhibitors, leukotriene receptor antagonists, antihistamines, opioid agonists (eg, ⁇ -opioid agonists), phosphodiesterase
- Non-steroidal anti-inflammatory drugs include, for example, azaline preparations such as Sazapyrine, sodium salicylate, aspirin, aspirin / dialuminate, diflunisal, indomethacin, suprofen, ufenamate, dimethylisopropylazulene, bufaxamac, felbinac, diclofenac, tolmetin sodium, Crinolyl, Fenbufen, Napemetone, Progouritacin, Indomethacin Farnesyl, Acemetacin, Progouritacin maleate, Ampenac sodium, Mofezolac, Etodolac, Ibuprofen, Ibuprofen piconol, Naproxen, Flurbiprofen, Flurbiprofen axetil, Ketoprofen , Phenoprofen calcium, thiaprofen, oxaprozin, pranop Fen, loxoprofen sodium, alumino
- steroid drugs include clobetasol propionate, diflorazone acetate, fluoconide, mometasone furanate, betamethasone dipropionate, betamethasone butyrate propionate, betamethasone valerate, difluprednate, pudesonide, diflucortron valerate.
- beclomethasone propionate As inhalants, beclomethasone propionate, fluticasone propionate, budesonide, flunisolide, triamcinolone, ST-126P, ciclesonide, dexamethasone paromithionate, mometasone furan carbonate, plasterone sulfonate, deflazacote, methylprednisolone sulphate, and methyl Examples include prednisolone sodium succinate.
- immunosuppressive drugs include protopic, tacrolimus (FK-506), methotrexate, cyclosporine, neoral, ascomycin, leflunomide, bucillamine, and salazosulfapyridine.
- Prostaglandins include PGE receptors (EP1, EP2, EP3, EP4), PGD receptors (DP, CRTH2), PGF receptors (FP), PGI receptors (IP ), Agonists such as TX receptor (TP), antagonists and the like.
- Antiallergic agents include, for example, amlexanox, azelastine hydrochloride, israpafant, ibudilast, imitrodast sodium, ebastine, epinastine hydrochloride, emedastine fumarate, oxatomide, ozagrel hydrochloride, olopatadine hydrochloride, cromoglycate, cromoglycate sodium, fumarate Ketotifen acid, seratrodast, cetirizine hydrochloride, suplatast tosylate, tazanolast, terfenadine, domitroban calcium hydrate, tranilast, nedocromil, fexofenadine, fexofenadine, pemirolast potassium, mequitazine, ramatroban, repirinast, loratadine, etc. Is mentioned.
- mediator release inhibitor examples include tranilast, sodium cromoglycate, amlexanox, repirinast, ibudilast, dazanolast, and pemirolast potassium.
- Leukotriene receptor antagonists include, for example, pranlukast hydrate, montelukast, zafirlukast, MCC-847, KCA-757, CS-615, YM-158, L-740515, CP-195494, LM-1484, RS -635, A-93178, S-36496, BIIL-284, ONO-4057, and the like.
- Antihistamines include, for example, ketotifen fumarate, mequitazine, azelastine hydrochloride, oxatomide, terfenadine, emedastine fumarate, epinastine hydrochloride, astemizole, ebastine, cetirizine hydrochloride, bepotastine, fexofenadine, loratadine, desloratadine, TA olopatadine hydrochloride, TA -427, ZCR-2060, NIP-530, mometasone furoate, mizolastine, BP-294, andlast, auranofin, and acribastine.
- opioid agonists examples include codeine, fentanyl, hydromorphone, levorphanol, meperidine, methadone, morphine, oxycodone, oxymorphone, propoxyphene, and the like, and ⁇ -opioid agonists include TRK-820, nalflavine, U50488H etc. are mentioned.
- Phosphodiesterase inhibitors include, for example, phosphodiesterase 4 inhibitors rolipram, silomilast, Bay19-8004, NIK-616, roflumilast (BY-217), cypamfilin (BRL-61063), atizolam (CP-80633), SCH-355991 YM-976, V-11294A, PD-168787, D-4396, and IC-485, and the phosphodiesterase 5 inhibitor includes, for example, sildenafil.
- Examples of the forskolin preparation include colforsin hydrochloride part, forsulin and the like.
- nitric oxide synthase inhibitors include N G -nitro-L-arginine methyl ester (L-NAME), N G -monomethyl-L-arginine (L-NMMA) and N G -nitro-L-arginine. (L-NNA) and the like.
- cannabinoid-2 receptor stimulants include compounds described in S-77469, S-444823, WO 02/010135 pamphlet, and WO 03/064389 pamphlet.
- Examples of the decoy preparation include NF- ⁇ B.
- chymase inhibitors examples include TPC-806, SUN13834 and the like.
- Examples of other drugs for complementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against ulcerative colitis include, for example, steroid drugs, immunosuppressive drugs, aminosalicylic acid preparations, chymase inhibitors, and other drugs. Examples include drugs.
- Examples of the steroid drug include the steroid drugs listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- immunosuppressive drug examples include those listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on atopic dermatitis.
- aminosalicylic acid preparations examples include pentasa, salazosulfapyridine, and mesalazine.
- chymase inhibitor examples include the chymase inhibitors listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- IL-6 antagonists such as anti-interleukin-6 (hereinafter abbreviated as IL-6) receptor antibodies. Can be mentioned.
- Examples of other drugs for complementing and / or enhancing the preventive and / or therapeutic effects of the compounds of the present invention on heart failure include, for example, diuretics, angiotensin converting enzyme inhibitors, angiotensin II receptor antagonists, antiarrhythmic drugs, digitalis Formulations, chymase inhibitors, renin inhibitors, calcium antagonists, beta blockers, nitrates, antialdosterone drugs, cardiotonic drugs, antiplatelet drugs, anticoagulants and the like.
- diuretics examples include hydrochlorothiazide, trichlormethiazide, benchylhydrochlorothiazide, indapamide, chlorthalidone, mefluzide, methiclan, trypamide, furosemide, torasemide, ethacrynic acid, bumetanide, piretanide, azosemide, triamterene and the like.
- angiotensin converting enzyme inhibitor examples include captopril, enalapril, alacepril, delapril, cilazapril, benazepril, lisinopril, and perindopril.
- angiotensin II receptor antagonist examples include candesartan, losartan, olmesartan, valsartan, telmisartan, irbesartan, candesartan cilexetil, losartan potassium, omersartan medoxomil, losartan potassium, losartan potassium hydrochlorothiazide, bropres plus, dioban HCT, valsartan HCTZ, CS-866CMB, VAA489, CS-8663, CS-866AZ, candelsartan amlodipine, telmisartan HCTZ, TAK-491, TAK-536, TAK-591, KT3-671 and the like.
- antiarrhythmic drug examples include amiodarone.
- Examples of digitalis preparations include digoxin.
- chymase inhibitor examples include the chymase inhibitors listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- renin inhibitor examples include aliskiren and aliskiren fumarate.
- calcium antagonists examples include nifedipine, nicardipine, nicardipine hydrochloride, nilvapidine, nisoldipine, nitrendipine, manidipine, manidipine hydrochloride, benidipine, varnidipine hydrochloride, varnidipine, amlodipine, amlodipine besylate, efonidipine hydrochloride, felodipine hydrochloride, felodipine, Examples include cilnidipine, alanidipine, azelnidipine, diltiazem, diltiazem hydrochloride, verapamil, verapamil hydrochloride, bepridil, bepridil hydrochloride, and the like.
- beta blocker examples include carvedilol, bisoprolol, metoprolol, propranolol, nadolol, nipradilol, chilysolol, pindolol, penbutolol, carteolol, bopindolol, atenolol, acebutolol, and ceriprolol.
- nitric acid drugs examples include nitroglycerin, amyl nitrite, and isosorbide nitrate.
- antialdosterone drug examples include spironolactone, eplerenone and the like.
- cardiotonic agent examples include pimobendan, denopamine, docarpamine, vesnarinone and the like.
- Antiplatelet agents include, for example, cyclooxygenase inhibitors, thromboxane synthase inhibitors, thromboxane receptor antagonists, adenosine-2-phosphate receptor antagonists, serotonin receptor antagonists, phosphodiesterase inhibitors, prostaglandins And GPIIb / IIIa antagonists.
- aspirin ticlopidine hydrochloride, clopidogrel sulfate, prasugrel, CS-747, ticagreler, AZD6140, erinogrel, PRT128, YM337, YM028, dipyridamole, cilostazol, beraprost sodium, sarpogrelate hydrochloride, sodium ozagrel, tivoxibane, Etc.
- anticoagulant examples include heparins (for example, heparin sodium, dalteparin sodium, heparin calcium, parnaparin sodium, leviparin sodium, etc.), heparin analogs (for example, danaparoid sodium, enoxaparin, nadroparin, bemiparin, Low molecular weight heparins such as reviparin and tinzaparin), thrombin inhibitors (for example, argatroban, ximelagatran, melagatran, dabigatran, bivalirdin (hirlog), repirudine, hirudin, decylzine, SSR-182289A, SR-123781A, S-18326, AZD -0837, LB-30870, L-375378, MCC-977, AT-1362, etc.), activated protein C preparations such as CTC-111, active dorotreco Alpha, antithrombin III preparations, gabexate mesylate
- Drug TTP-889, IXa aptamer (RB006), activated blood coagulation factor IX inhibitor such as 224AE3, thrombomodulin preparation such as ART-123, sodium citrate, carboxypeptida such as AZD-9684 Ze U (TAFI) inhibitor, etc.), vitamin K antagonists (e.g., warfarin, Coumadin, warfarin, etc.) and the like.
- Examples of other drugs for complementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on pulmonary fibrosis include, for example, steroid drugs, immunosuppressive drugs, and antifibrotic drugs.
- Examples of the steroid drug include the steroid drugs listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- immunosuppressive drug examples include those listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on atopic dermatitis.
- antifibrotic agent examples include pirfenidone, pyretha, chymase inhibitor (for example, TPC-806, SUN13834, etc.) and the like.
- Examples of other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compounds of the present invention on aneurysms include, for example, hypoglycemic drugs, antihypertensive drugs, lipid improving drugs, anti-obesity drugs and the like.
- hypoglycemic agents include repaglinide, miglitol, exenatide, nateglinide, mitiglinide calcium hydrate, pioglitazone hydrochloride, rosiglitazone maleate, balaglitazone, R-483, netoglitazole, naveglitazar, T-131, T-131 E7001, CLX-0921, metformin, buformin, miglitol, voglibose, acarbose, insulinotropin, liraglutide, CJC-1131, GLP-1, R-1583, LY-307161, rGLP-1 (Betatropin), sitagliptin, vildagliptin LAF-237, P-32 / 98, P-93 / 01, TS-021, 815541, 82596 , 823093, TA-6666, and MK-0431, and the like.
- antihypertensive drugs examples include captopril, enalapril, alacepril, delapril, cilazapril, benazepril, lisinopril, perindopril, candesartan, losartan, olmesartan, valsartan, telmisartan, irbesartan, candesartan cilexetil, losartan potassium, omsartan doxanol, Losartan potassium hydrochlorothiazide, Blopress plus, Dioban HCT, Valsartan HCTZ, CS-866CMB, VAA489, CS-8663, CS-866AZ, Kandelsartan amlodipine, Telmisartan HCTZ, TAK-491, TAK-536, TAK-591 KT3-671, doxazosin, urapidil, terazosin, prazosin, etc. Can be mentioned.
- anti-obesity agents examples include rimonabant, mazindol, orlistat, sibutramine and the like.
- lipid improvers examples include atorvastatin, simvastatin, pitavastatin, pravastatin, fluvastatin, lovastatin, ezetimibe, bezafibrate, clinofibrate, clofibrate aluminum, fenofibrate, clofibrate, probucol, and eicosapentaenoic acid.
- nonalcoholic steatohepatitis examples include, for example, hypoglycemic drugs, antihypertensive drugs, lipid-improving drugs, Examples include obesity drugs, liver protection drugs, antioxidant drugs, and antifibrotic drugs.
- hypoglycemic drug examples include hypoglycemic drugs listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on aneurysms.
- antihypertensive agent examples include antihypertensive agents listed as other agents for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on aneurysms.
- anti-obesity drugs examples include the anti-obesity drugs listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on aneurysms.
- lipid-improving drug examples include lipid-improving drugs listed as other drugs for complementation and / or enhancement of the preventive and / or therapeutic effect of the compound of the present invention on aneurysms.
- liver protectants include strong neominophagen C, ursodeoxycholic acid, Sho-saiko-to and the like.
- antioxidants examples include vitamin C, vitamin E, N-acetylcysteine, betaine, and s-adenosylmethionine.
- anti-fibrotic drug examples include the anti-fibrotic drugs listed as other drugs for complementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on pulmonary fibrosis.
- agents for complementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on non-steroidal anti-inflammatory analgesic-induced peptic ulcer include, for example, prostaglandin preparations, H2 receptor antagonists, Examples include proton pump inhibitors.
- prostaglandin preparations include misoprostol, cytotech, and cobiprostone.
- H2 receptor antagonist examples include cimetidine, tagamet, ranitidine hydrochloride, zantac, famotidine, gaster, nizatidine, asinone, loxatidine acetate hydrochloride, altertate, lafutidine, protecazine, stagger, and the like.
- proton pump inhibitors examples include omepral, omeprazone, takepron, pariet, lansoprazole, omeprazole, rabeprazole and the like.
- Examples of other drugs for complementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention on allergic conjunctivitis include antiallergic drugs and steroid drugs.
- antiallergic agent examples include the antiallergic agents listed as other agents for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- Examples of the steroid drug include the steroid drugs listed as other drugs for supplementing and / or enhancing the preventive and / or therapeutic effect of the compound of the present invention against atopic dermatitis.
- the mass ratio between the compound of the present invention and other drugs is not particularly limited.
- Other drugs may be administered in combination of any two or more.
- drugs that complement and / or enhance the preventive and / or therapeutic effects of the compounds of the present invention include not only those that have been found so far, but also those that will be found in the future based on the above-mentioned mechanism. It is.
- the compound of the present invention or the concomitant agent of the compound of the present invention and another drug for the above purpose it is usually administered systemically or locally in an oral or parenteral form.
- the dose varies depending on age, body weight, symptoms, therapeutic effect, administration method, treatment time, etc., but is usually orally administered once to several times a day in the range of 1 mg to 1000 mg per adult. Or administered parenterally (preferably nasal drops, eye drops or ointments) once to several times per day in the range of 0.1 mg to 100 mg per adult, or It is continuously administered intravenously in the range of 1 to 24 hours per day.
- a solid preparation for internal use for oral administration When administering the compound of the present invention, or a combination of the compound of the present invention and another drug, a solid preparation for internal use for oral administration, a liquid for internal use, and an injection, a preparation for external use, a sitting for parenteral administration. It is used as an agent, eye drops, inhalant and the like.
- Oral solid preparations for oral administration include tablets, pills, capsules, powders, granules and the like.
- Capsules include hard capsules and soft capsules.
- Tablets include sublingual tablets, buccal adhesive tablets, buccal quick disintegrating tablets and the like.
- one or more active substances are left as they are, or excipients (lactose, mannitol, glucose, microcrystalline cellulose, starch, etc.), binders (hydroxylpropylcellulose, polyvinylpyrrolidone, Mixed with magnesium metasilicate aluminate, etc.), disintegrating agents (such as calcium calcium glycolate), lubricants (such as magnesium stearate), stabilizers, solubilizing agents (such as glutamic acid, aspartic acid), etc. Used by formulating.
- excipients lactose, mannitol, glucose, microcrystalline cellulose, starch, etc.
- binders hydroxylpropylcellulose, polyvinylpyrrolidone, Mixed with magnesium metasilicate aluminate, etc.
- disintegrating agents such as calcium calcium glycolate
- lubricants such as magnesium stearate
- solubilizing agents such as glutamic acid, aspartic acid
- a coating agent sucrose, gelatin, hydroxylpropylcellulose, hydroxylpropylmethylcellulose phthalate, etc.
- a coating agent sucrose, gelatin, hydroxylpropylcellulose, hydroxylpropylmethylcellulose phthalate, etc.
- capsules of absorbable substances such as gelatin.
- Oral solutions for oral administration include pharmaceutically acceptable solutions, suspensions, emulsions, syrups, elixirs and the like.
- one or more active substances are dissolved, suspended or emulsified in a commonly used diluent (purified water, ethanol or a mixture thereof).
- this liquid agent may contain a wetting agent, a suspending agent, an emulsifier, a sweetening agent, a flavoring agent, a fragrance, a preservative, a buffering agent and the like.
- External dosage forms for parenteral administration include, for example, ointments, gels, creams, poultices, patches, liniments, sprays, inhalants, sprays, aerosols, eye drops, and Includes nasal drops and the like. These contain one or more active substances and are prepared by known methods or commonly used formulations.
- Sprays, inhalants and sprays are buffers that provide isotonicity with stabilizers such as sodium bisulfite in addition to commonly used diluents, such as sodium chloride, sodium citrate or citric acid.
- stabilizers such as sodium bisulfite
- An isotonic agent may be contained.
- a method for producing a spray is described in detail in, for example, US Pat. Nos. 2,868,691 and 3,095,355.
- injections for parenteral administration are solutions, suspensions, emulsions and solid injections used by dissolving or suspending in a solvent at the time of use.
- An injection is used by dissolving, suspending or emulsifying one or more active substances in a solvent.
- the solvent for example, distilled water for injection, physiological saline, vegetable oil, propylene glycol, polyethylene glycol, alcohols such as ethanol, and combinations thereof are used.
- this injection may contain a stabilizer, a solubilizer (glutamic acid, aspartic acid, polysorbate 80 (registered trademark), etc.), a suspending agent, an emulsifier, a soothing agent, a buffering agent, a preservative, and the like.
- a sterile solid preparation for example, a lyophilized product, can be produced and used by dissolving it in sterilized or sterile distilled water for injection or other solvent before use.
- compositions for parenteral administration include suppositories for rectal administration and pessaries for intravaginal administration, which contain one or more active substances and are prescribed by conventional methods.
- the location of separation by chromatography and the solvent in parentheses shown in TLC indicate the elution solvent or developing solvent used, and the ratio indicates the volume ratio.
- the solvent in parentheses shown in the NMR part indicates the solvent used for the measurement.
- Example 1 Methyl 2-acetyl-1 -naphthoate Under an argon atmosphere, methyl 2-trifluoromethanesulfonyloxy-1-naphthoate (3.34 g) in acetonitrile (20 mL) was added to butyl vinyl ether (6.4 mL), palladium acetate. (0.17 g), triphenylphosphine (0.39 g), and triethylamine (1.7 mL) were added, and the mixture was stirred overnight under reflux. The reaction mixture was filtered through Celite (registered trademark), and the filtrate was concentrated under reduced pressure. The obtained residue was dissolved in tetrahydrofuran (200 mL) and ice-cooled.
- Example 4 Methyl 2- (3-formylimidazo [1,2-a] pyridin-2-yl) -1-naphthoate
- the compound prepared in Example 3 (172 mg) was converted to phosphorus oxychloride (1.7 mL). Suspended and stirred at room temperature. Dimethylformamide (0.046 mL) was added and stirred at 70 ° C. for 5.5 hours. The reaction mixture was ice-cooled, neutralized with 2M aqueous sodium hydroxide solution, and extracted with ethyl acetate. The obtained organic layer was washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to give the title compound (196 mg) having the following physical data.
- Example 5 Methyl 2- [3- (hydroxymethyl) imidazo [1,2-a] pyridin-2-yl] -1-naphthoate
- the compound prepared in Example 4 (195 mg) was added to tetrahydrofuran (1.5 mL).
- a solution of sodium borohydride (34 mg) in water (0.4 mL) was added under ice cooling and stirred for 1 hour.
- a saturated aqueous ammonium chloride solution was added to the reaction mixture, and the mixture was extracted twice with ethyl acetate. The obtained organic layers were combined, washed successively with water, saturated aqueous sodium hydrogen carbonate solution and saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure.
- Example 6 Methyl 2- ⁇ 3-[(4-Methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoate Prepared in Example 5 The compound (47 mg) and 4-methylindole (20 mg) were suspended in water (0.25 mL) and stirred at room temperature. Acetic acid (0.024 mL) was added and stirred at 95 ° C. overnight. A saturated aqueous sodium hydrogen carbonate solution was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained organic layer was washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure.
- Example 7 2- ⁇ 3-[(4-Methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoic acid
- Example 8 Methyl 2- acetylbenzoate Under ice-cooling, methyl iodide (66.85 g) and potassium carbonate (118.40 g) were added to a solution of 2-acetylbenzoic acid (70.29 g) in dimethylformamide (500 mL). Stir for 1 hour. The reaction mixture was filtered through Celite (registered trademark), and the filtrate was concentrated under reduced pressure. A saturated aqueous ammonium chloride solution was added to the resulting residue, and the mixture was extracted with ethyl acetate.
- Example 9 Methyl 2- [3- (hydroxymethyl) -6-methylimidazo [1,2-a] pyridin-2-yl] benzoate Using 5-methyl-2-aminopyridine instead of 2-aminopyridine Using the compound produced in Example 8 instead of the compound produced in Example 1, the same physical properties as in Example 2 ⁇ Example 3 ⁇ Example 4 ⁇ Example 5 were obtained. The title compound was obtained.
- Example 10 To a solution of 1,4-dimethyl-1H-indole 4-methylindole (22.08 g) in dimethylformamide (200 mL) was added methyl iodide (28.44 g) and cesium carbonate (82.26 g) Stir at room temperature overnight. The reaction mixture was filtered through Celite (registered trademark), and the filtrate was concentrated under reduced pressure. A saturated aqueous ammonium chloride solution was added to the resulting residue, and the mixture was extracted with ethyl acetate. The obtained organic layer was washed with water and saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure.
- methyl iodide 28.44 g
- cesium carbonate 82.26 g
- Example 11 4-Methyl 2- ⁇ 3-[(1,4-dimethyl-1H-indol-3-yl) methyl] -6-methylimidazo [1,2-a] pyridin-2-yl ⁇ benzoate
- Example 10 4-Methyl 2- ⁇ 3-[(1,4-dimethyl-1H-indol-3-yl) methyl] -6-methylimidazo [1,2-a] pyridin-2-yl ⁇ benzoate
- Example 13 2- ⁇ 3-[(1,4-Dimethyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid Compound prepared in Example 5 Instead of using the compound prepared in Example 12 and using the compound prepared in Example 10 instead of 4-methylindole, the same procedure as in Example 6 ⁇ Example 7 was performed, and the following procedure was performed. The compound of the present invention having physical property values was obtained.
- Example 14 1-ethyl-4-methyl-1H-indole
- the title compound having the following physical data was obtained by conducting the same operation as in Example 10 using ethyl iodide instead of methyl iodide. It was.
- Example 15 2- ⁇ 3-[(1-Ethyl-4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid 4-methylindole
- Example 15 2- ⁇ 3-[(1-Ethyl-4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid 4-methylindole
- Example 16 2- ⁇ 6-Fluoro-3-[(4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoic acid 2- Using 5-fluoro-2-aminopyridine instead of aminopyridine, the same physical properties as in Example 3 ⁇ Example 4 ⁇ Example 5 ⁇ Example 6 ⁇ Example 7 were obtained. The compound of the present invention was obtained.
- Example 17 7-Chloro-5-fluoro-4-methyl-1H-indole
- 4-chloro-2-fluoro-5-nitrotoluene (1.9 g) was dissolved in anhydrous tetrahydrofuran (100 mL). Stir at 50 ° C.
- 40 mL of vinylmagnesium bromide (1.0 M tetrahydrofuran solution) was added dropwise. The reaction mixture was gradually warmed to ⁇ 20 ° C. over 40 minutes.
- a saturated aqueous ammonium chloride solution was added to the reaction mixture, and the mixture was extracted with ethyl acetate.
- Example 18 5-Fluoro-4-methyl-1H-indole
- the compound prepared in Example 17 500 mg was dissolved in ethanol (9 mL), triethylamine (0.46 mL) was added, and the mixture was stirred at room temperature. Under an argon atmosphere, 50 mg of 10% Pd—C (50% wet) was added. The reaction mixture was changed from an argon atmosphere to a hydrogen atmosphere and then stirred for 4.5 hours. The reaction mixture was filtered through Celite (registered trademark), and the filtrate was concentrated under reduced pressure.
- Example 19 2- ⁇ 3-[(5-Fluoro-4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid 4-methylindole
- Example 19 2- ⁇ 3-[(5-Fluoro-4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid 4-methylindole
- Example 20 2- ⁇ 3-[(4-Methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoic acid hydrochloride
- Example 7 The prepared compound (190 mg) was dissolved in 4N hydrochloric acid / 1,4-dioxane solution (9 mL) and stirred at room temperature for 5 minutes. Thereafter, the reaction mixture was concentrated under reduced pressure. The obtained residue was dissolved in methanol and concentrated again under reduced pressure to obtain the compound of the present invention having the following physical properties.
- Example 21 2- ⁇ 3-[(4-Methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoic acid sodium salt
- Example 7 To a solution of the produced compound (5.0 g) in ethanol (100 mL) / water (50 mL) was added 1N aqueous sodium hydroxide solution (11.6 mL), and the mixture was refluxed at 80 ° C. The reaction mixture was allowed to cool to room temperature and then concentrated under reduced pressure. Water (30 mL) and ethanol (10 mL) were added to the obtained residue and refluxed. The reaction mixture was allowed to cool to room temperature, and the precipitated crystals were filtered.
- Example 22 2- ⁇ 3-[(1,4-Dimethyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ benzoic acid sodium salt
- Example 13 Using the obtained compound, the same operation as in Example 21 was performed to obtain the compound of the present invention having the following physical property values.
- Example 23- (1) to (4) Using the corresponding alkyl halide instead of methyl iodide, the same operation as in Example 10 ⁇ Example 7 ⁇ Example 20 was performed to obtain the compound of the present invention having the following physical property values (however, Example 23- (4) was obtained by carrying out the same operation as in Example 10 ⁇ Example 7.
- Example 23- (1): 2- (3- ⁇ [4-Methyl-1- (2-propanyl) -1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridin-2-yl ) -1-Naphthoic acid hydrochloride TLC: Rf 0.35 (methylene chloride: methanol 9: 1); 1 H-NMR (DMSO-D 6 ): ⁇ 1.10 (d, 6 H), 2.68 (s, 3 H), 4.31-4.60 (m, 1 H), 4.76 (s, 2 H), 6.48 (s, 1 H), 6.72 (d, 1 H), 6.84-7.02 (m, 2 H), 7.18 (d, 1 H), 7.24-7.41 (m, 1 H), 7.48-7.73 (m, 4 H), 7.86-8.12 (m, 3 H), 8.25 (d, 1 H).
- Example 24 2- ⁇ 3-[(5-Fluoro-4-methyl-1H-indol-3-yl) methyl] imidazo [1,2-a] pyridin-2-yl ⁇ -1-naphthoic acid 4- Using the compound produced in Example 18 instead of methylindole, the same operation as in Example 6 ⁇ Example 7 was performed to obtain the compound of the present invention having the following physical property values.
- Example 25 Methyl 2- [3-( ⁇ 4-methyl-1- [2- (tetrahydro-2H-pyran-2-yloxy) ethyl] -1H-indol-3-yl ⁇ methyl) imidazo [1,2 -A] pyridin-2-yl] -1-naphthoate
- 2- (2-bromoethoxy) tetrahydro-2H-pyran instead of methyl iodide.
- the title compound having the following physical property values was obtained.
- Example 26 Methyl 2- (3- ⁇ [1- (2-hydroxyethyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridin-2-yl)- 1-Naphthoate
- the compound prepared in Example 25 (390 mg) was dissolved in 12 mL of acetic acid and 3 mL of water and stirred at 80 ° C. for 1.5 hours.
- the reaction mixture was cooled to room temperature, neutralized with 2N aqueous sodium hydroxide solution, and extracted with ethyl acetate.
- the obtained organic layer was washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure.
- Example 27 2- (3- ⁇ [1- (2-hydroxyethyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridin-2-yl) -1 -Naphthoic acid hydrochloride Using the compound prepared in Example 26, the same operation as in Example 7 ⁇ Example 20 was performed to obtain the compound of the present invention having the following physical property values.
- Example 28 Methyl 2- [3-( ⁇ 1- [3- (dimethylamino) -2-hydroxypropyl] -4-methyl-1H-indol-3-yl ⁇ methyl) imidazo [1,2-a] Pyridin-2-yl] -1-naphthoate Under ice-cooling, 60% strength sodium hydride (16 mg) was added to a solution of the compound prepared in Example 6 (150 mg) in dimethylformamide (3.4 mL). Added and stirred for several minutes. Under ice cooling, epibromohydrin (35 ⁇ L) was added to the reaction mixture and stirred for 30 minutes.
- Example 29 2- [3-( ⁇ 1- [3- (Dimethylamino) -2-hydroxypropyl] -4-methyl-1H-indol-3-yl ⁇ methyl) imidazo [1,2-a] pyridine -2-yl] -1-naphthoic acid sodium salt Using the compound produced in Example 28, the same procedure as in Example 7 ⁇ Example 21 was performed, and the present invention having the following physical properties A compound was obtained.
- Example 30 Methyl 2- (3- ⁇ [1- (2,3-dihydroxypropyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridin-2-yl ) -1-Naphthoate
- 60% strength sodium hydride (16 mg) was added at room temperature and stirred for several minutes.
- glycidol 0.1 mL was added to the reaction mixture, and the mixture was stirred at 80 ° C. for 30 minutes. Water was added to the reaction mixture, and the mixture was extracted with ethyl acetate.
- Example 31 2- (3- ⁇ [1- (2,3-dihydroxypropyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridin-2-yl) 1-Naphthoic acid hydrochloride
- the compound of the present invention having the following physical data was obtained by the same procedures as in Example 7 ⁇ Example 20 using the compound produced in Example 30.
- Example 32- (1) to (47) Using the corresponding alkyl halide in place of methyl iodide, the same operation as in Example 10 ⁇ Example 7 was performed to obtain the compound of the present invention having the following physical property values.
- Example 32- (14): 2- (3- ⁇ [1- (3-hydroxypropyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] pyridine-2- Yl) -1-naphthoic acid TLC: Rf 0.23 (chloroform: methanol: aqueous ammonia 79: 20: 1); 1 H-NMR (CDCl 3 ): ⁇ 1.63-1.69 (m, 2H), 2.64 (s, 3H), 3.23 (t, 2H), 3.92 (t, 2H), 4.69 (s, 2H), 6.42 (s , 1H), 6.71 (d, 1H), 6.80-6.83 (m, 1H), 6.95 (dd, 1H), 7.13 (d, 1H), 7.19-7.23 (m, 1H), 7.49-7.59 (m, 4H ), 7.84 (d, 1H), 7.88 (d, 1H), 8.05-8.09 (m, 2H).
- Example 32- (15): 2- [3-( ⁇ 1- [2- (dipropan-2-ylamino) ethyl] -4-methyl-1H-indol-3-yl ⁇ methyl) imidazo [1,2- a] pyridin-2-yl] -1-naphthoic acid
- Example 32- (18): 2- [3-( ⁇ 1- [2- (1,1-dioxidethiomorpholin-4-yl) ethyl] -4-methyl-1H-indol-3-yl ⁇ methyl ) Imidazo [1,2-a] pyridin-2-yl] -1-naphthoic acid TLC: Rf 0.26 (chloroform: methanol: aqueous ammonia 79: 20: 1); 1 H-NMR (DMSO-D 6 ): ⁇ 2.63 (t, 2H), 2.66 (s, 3H), 2.71-2.78 (m, 8H), 3.95 (t, 2H), 4.71 (s, 2H), 6.46 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.94-6.98 (m, 1H), 7.15 (d, 1H), 7.20-7.24 (m, 1H), 7.49-7.59 (m, 4H), 7.86 (d, 1H), 7.
- 1 H-NMR (CDCl 3) ⁇ 2.67 (s, 3H), 2.95 (t, 2H), 4.13 (t, 2H), 4.55 (s, 2H), 6.07 (s, 1H), 6.65 (d, 1H ), 6.77-6.83 (m, 4H), 7.01 (d, 1H), 7.06-7.14 (m, 3H), 7.27-7.38 (m, 3H), 7.54 (d, 1H), 7.68 (d, 1H), 7.73 (d, 1H), 7.89 (d, 1H), 8.15 (d, 1H).
- Example 32- (27): 2- ⁇ 3-[(4-methyl-1- ⁇ 2- [methyl (phenyl) amino] ethyl ⁇ -1H-indol-3-yl) methyl] imidazo [1,2- a] Pyridin-2-yl ⁇ -1-naphthoic acid TLC: Rf 0.66 (chloroform: methanol: aqueous ammonia 79: 20: 1); 1 H-NMR (DMSO-D 6 ): ⁇ 2.48 (s, 3H), 2.65 (s, 3H), 3.42 (t, 2H), 4.04 (t, 2H), 4.69 (s, 2H), 6.37 (s , 1H), 6.45 (d, 2H), 6.53-6.57 (m, 1H), 6.73 (d, 1H), 6.79-6.82 (m, 1H), 6.92-6.96 (m, 1H), 7.00-7.07 (m 3H), 7.21-7.25 (m, 1H), 7.49-7.59 (m,
- Example 32- (35): 2- (3- ⁇ [1- (but-3-en-1-yl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a ] Pyridin-2-yl) -1-naphthoic acid TLC: Rf 0.63 (chloroform: methanol: aqueous ammonia 79: 20: 1); 1 H-NMR (DMSO-D 6 ): ⁇ 2.23-2.29 (m, 2H), 2.64 (s, 3H), 3.92 (t, 2H), 4.69 (s, 2H), 4.77-4.85 (m, 2H) , 5.50-5.61 (m, 1H), 6.40 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.94-6.98 (m, 1H), 7.13 (d, 1H), 7.22 -7.26 (m, 1H), 7.51-7.59 (m, 4H), 7.87-7.92 (m, 2
- 1 H-NMR (CDCl 3) ⁇ 1.00 (t, 6H), 2.68-3.00 (m, 9H), 4.08-4.10 (m, 2H), 4.51 (s, 2H), 6.61 (s, 1H), 6.69 -6.72 (m, 1H), 6.79-6.81 (m, 1H), 7.04-7.05 (m, 2H), 7.13-7.18 (m, 1H), 7.38-7.50 (m, 3H), 7.68 (d, 1H) , 7.76-7.80 (m, 2H), 7.85 (d, 1H), 8.19
- 1 H-NMR (DMSO-D 6) ⁇ 2.64 (s, 3H), 3.06-3.09 (m, 5H), 3.72-3.82 (m, 2H), 3.91-3.96 (m, 1H), 4.69 (s, 2H), 6.43 (s, 1H), 6.70 (d, 1H), 6.78-6.81 (m, 1H), 6.92-6.95 (m, 1H), 7.13 (d, 1H), 7.18-7.22 (m, 1H) 7.45-7.58 (m, 4H), 7.78 (d, 1H), 7.85 (d, 1H), 8.06-8
- Example 33- (3): 2- (3- ⁇ [1- (2-hydroxy-2-methylpropyl) -4-methyl-1H-indol-3-yl] methyl ⁇ imidazo [1,2-a] Pyridin-2-yl) -1-naphthoic acid TLC: Rf 0.49 (chloroform: methanol: aqueous ammonia 79: 20: 1); 1 H-NMR (DMSO-D 6 ): ⁇ 0.93 (s, 6H), 2.64 (s, 3H), 3.78 (s, 2H), 4.70 (s, 2H), 6.43 (s, 1H), 6.70 (d , 1H), 6.80-6.83 (m, 1H), 6.92-6.95 (m, 1H), 7.21-7.25 (m, 2H), 7.51-7.61 (m, 4H), 7.88-7.93 (m, 2H), 8.02 (d, 1H), 8,11 (d, 1H).
- Pharmacological Experiment 1 Measurement of human chymase inhibitory activity Human Chymase Pure (hereinafter, .East Coast Biologics Inc. abbreviated as chymase) is stored at -20 ° C. freezer was used by diluting with distilled water immediately prior to use .
- N-Succinyl-Ala-Ala-Pro-Phe-p-nitronide hereinafter abbreviated as peptide substrate; Sigma-Aldrich
- DMSO dimethyl sulfoxide
- the compounds of the present invention and comparative compounds were made into 10 mmol / L solutions in DMSO and stored in a freezer at ⁇ 20 ° C. On the day of the measurement, serial dilution was performed with 50% DMSO so that the concentration was 10 times the target final concentration to prepare a compound solution of the present invention and a comparative compound solution.
- Peptide substrate solution (final) diluted in 96-well microplate with 10 ⁇ L of the compound solution of the present invention and comparative compound solution and assay buffer (50 mmol / L Tris-HCl, 150 mmol / L NaCl, 50 units / mL heparin (pH 7.6)) 80 ⁇ L of a concentration of 2.5 mmol / L) was added and incubated at 37 ° C. for 10 minutes.
- 10 ⁇ L of chymase was added to a final concentration of 0.1 ⁇ g / L, and the enzyme reaction was started. Absorbance at 405 nm was monitored at 30 second intervals for 5 minutes, and the reaction rate ( ⁇ mO.D. / min) was measured.
- the treatment concentrations of the compound of the present invention and the comparative compound were 0, 6.25, 12.5, 25, 50 and 100 ⁇ mol / L.
- the ATP concentration in the cells was measured using Celltiter Glo luminescent assay kit (Promega), and the cytotoxic effects of the compounds of the present invention and comparative compounds were evaluated.
- the assay buffer of the measurement kit lysed the cells, and the concentration of ATP released from the cells was measured using luciferin-luciferase enzyme activity as an index. Luminescence was measured with a SpectraMax plate reader (Molecular device).
- the cytotoxic effect of the compound of the present invention and the comparative compound was expressed as a concentration (IC 10 and IC 50 ) that suppresses luminescence by 10% and 50%. That is, IC 10 value was calculated as the concentration that caused toxicity to 10% of cells and HepG2 cells, and IC 50 value was calculated as the concentration that caused toxicity to 50% of cells.
- the ratio of HepG2 cytotoxicity (described in Table 2) to human chymase inhibitory activity (described in Table 1) of the compounds of the present invention (IC 50 (HepG2 cytotoxicity) / IC 50 (human chymase inhibitory activity) ), IC 10 (HepG2 cytotoxicity) / IC 90 (human chymase inhibitory activity)) was very large compared to any of the comparative compounds. Therefore, the compound of the present invention can be a drug with a greatly reduced risk of hepatotoxicity when used as a preventive and / or therapeutic agent for chymase-mediated diseases.
- Pharmacological experiment example 3 Stability test on human liver microsome 5 ⁇ L of the compound of the present invention and a comparative compound (10 mmol / L DMSO solution) were diluted with 195 ⁇ L of 50% acetonitrile aqueous solution, and 250 ⁇ mol / L of the compound solution of the present invention and comparison A compound solution was made.
- NADPH Cofactor NADPH Regenerating System Solution A (BD Bioscience) and NADPH Regenerating System Solution B (BD Bioscience) 0.1 mol / L pH 7.4 phosphate buffer in a reaction vessel preheated to 37 ° C. in a water bath.
- Formulation Example 1 The following components were mixed by a conventional method and then tableted to obtain 10,000 tablets each containing 10 mg of the active ingredient.
- Formulation Example 2 The following components were mixed by a conventional method, filtered through a dust removal filter, filled in 5 ml aliquots, and sterilized by heating in an autoclave to obtain 10,000 ampoules containing 20 mg of active ingredient per ampoule.
- the compounds of the present invention have chymase inhibitory activity, and prevent and / or prevent chymase-mediated diseases such as skin diseases, cardiovascular diseases, gastrointestinal diseases, respiratory diseases, liver diseases, or eye diseases. It is effective for treatment.
Abstract
Description
Aaは、ハロゲン、C1-6直鎖状または分枝状アルキル、C1-6直鎖状または分枝状アルコキシ、C1-6直鎖状または分枝状アルキルチオ、C1-6直鎖状または分枝状アルキルスルホニル、フェニルおよびオキソから選択される1~3個の置換基で置換されていてもよい1個以上の-O-、-S-、-SO-、-SO2-および-NRaa-によって任意に組み込まれていてもよいC1-7直鎖状、分枝状または環状アルキル;
Eaは、-COORea等;
Reaは、それぞれ独立して、水素原子、C1-4アルキルまたはアリール;
Gaは、1個以上の-O-、-S-、-SO2-および-NRaa-によって任意に組み込まれていてもよいC1-6直鎖状または分枝状アルキル;
Maは、メチレン基、酸素原子、-N(Rba)-、-S(O)m a-;
Jaは、4~10個の炭素原子を有する、1つまたは複数の置換基を有していてもよい炭素環式芳香族化合物、または環内に酸素原子、窒素原子および硫黄原子から選択される1つ以上のヘテロ原子を含む複素環式芳香族化合物;
基の説明は、必要な部分を抜粋した。)で示される化合物またはその薬学的に許容される塩が、キマーゼ阻害剤である旨の記載がなされている。
Abは、置換もしくは無置換の炭素数6~11のアリーレン基、または置換もしくは無置換の酸素、窒素および硫黄原子を一つもしくは複数個環上に含んでいてもよい炭素数4~10のヘテロアリーレン基;
Ebは、-COOR3b等;
R3bは、水素原子または直鎖もしくは分枝状の炭素数1~6のアルキル基;
Gbは、置換もしくは無置換の炭素数1~6の直鎖もしくは分枝状のアルキレン基;
Mbは、単結合またはS(O)m b;
mbは、0~2の整数;
Jbは、置換もしくは無置換の炭素数6~11のアリール基あるいは置換もしくは無置換の酸素、窒素および硫黄原子を一つあるいは複数個環上に含んでいてもよい炭素数4~10のヘテロアリール基等;
Xbは、CHまたは窒素原子;
基の説明は、必要な部分を抜粋した。)で表されるベンズイミダゾール誘導体またはその医学上許容される塩が、キマーゼ阻害剤である旨の記載がなされている。
さらに、特許文献3には、一般式(Ic)
R1c及びR2cは、同時に又はそれぞれ独立に、水素原子、ハロゲン原子、トリハロメチル基、炭素数1~4のアルキル基等;
Acは、置換もしくは無置換の炭素数6~11のアリーレン基、又は置換もしくは無置換の酸素原子、窒素原子および硫黄原子からなる群から選ばれた一つ以上のヘテロ原子を環上に有する炭素数4~10のヘテロアリーレン基等;
Ecは、-COOR3c等;
R3cは、水素原子又は直鎖もしくは分枝状の炭素数1~6のアルキル基;
Gcは、置換又は無置換の炭素数1~6の直鎖又は分枝状のアルキレン基等;
Mcは、単結合又は-S(O)m c-;
m cは、0~2の整数;
Jcは、Mcが単結合の場合、置換もしくは無置換の炭素数6~11のアリール基、或いは置換もしくは無置換の酸素原子、窒素原子および硫黄原子からなる群から選ばれた一つ以上のヘテロ原子を環上に有する炭素数4~10のヘテロアリール基等;
Xcは、-CH=又は窒素原子;
Ycは、-CBc=又は窒素原子;
Zcは、炭素原子又は窒素原子;
Qcは、炭素原子又は窒素原子;
基の説明は、必要な部分を抜粋した。)で表される含窒素芳香環誘導体又はその塩は、URAT1阻害剤であり、該発明化合物はキマーゼ阻害活性を有している旨の記載がなされている。
〔1〕 一般式(I)
R1は、水素原子、メチル基、トリフルオロメチル基、ジフルオロメチル基、フッ素原子、または塩素原子を表し、
R2は、水素原子、1~6個のR11で置換されていてもよいC1~10のアルキル基、1~6個のR11で置換されていてもよいC2~10のアルケニル基、または1~6個のR11で置換されていてもよいC2~10のアルキニル基を表し、
R11は、
(i)ハロゲン原子、
(ii)OR8(基中、R8は
(1)水素原子、
(2)C1~3のアルキル基、
(3)C1~3のハロアルキル基、
(4)C5~6の炭素環基、
(5)1~2個の窒素原子、1個の酸素原子および/または酸化されていてもよい1個の硫黄原子を含有する5~6員のヘテロ環基、または
(6)水酸基、C1~3のアルコキシ基、またはNR9R10で置換されたC1~3のアルキル基、(基中、R9およびR10はそれぞれ独立して、水素原子、C1~3のアルキル基、またはフェニル基を表す。)、
(iii)NR9R10(基中、R9およびR10はそれぞれ独立して、水素原子、C1~3のアルキル基、またはフェニル基を表す。)、
(iv)ハロゲン原子、オキソ基、およびメチル基から選ばれる少なくとも1種の基で置換されていてもよいC5~6の炭素環基、または
(v)ハロゲン原子、オキソ基、およびメチル基から選ばれる少なくとも1種の基で置換されていてもよい、1~4個の窒素原子、1~2個の酸素原子および/または酸化されていてもよい1~2個の硫黄原子を含有する3~10員のへテロ環基を表し、
R3は、水素原子、メチル基、またはフッ素原子を表し、
R4は、水素原子、C1~2のアルキル基、C1~2のハロアルキル基、またはハロゲン原子を表し、
R5は、水素原子、C1~3のアルキル基、C1~3のハロアルキル基、またはハロゲン原子を表し、
R6は、水素原子、C1~3のアルキル基、C1~3のハロアルキル基、またはハロゲン原子を表し、
R7は、水素原子、C1~4のアルキル基、アリル基、トリクロロエチル基、ベンジル基、フェナシル基、p-メトキシベンジル基、トリチル基、または2-クロロトリチル基を表し、
pは、1~3の整数を表し、
qは、1~4の整数を表し、
rは、1~6の整数を表す。
ただし、p、q、およびrが、それぞれ2以上の整数を表す場合、R4、R5、およびR6は、それぞれ独立して、同一でも異なっていてもよい。)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグ、
〔2〕 一般式(I-a)
〔3〕
〔4〕 R2が、水素原子、1~6個のR12で置換されていてもよいC1~10のアルキル基、1~6個のR12で置換されていてもよいC2~10のアルケニル基、または1~6個のR12で置換されていてもよいC2~10のアルキニル基を表し、
R12が、ハロゲン原子、水酸基、ジメチルアミノ基、ピロリジニル基、N-メチルピロリジニル基、ピぺリジニル基、ハロゲン原子で置換されていてもよいフェノキシ基、ハロゲン原子で置換されていてもよいフェニル基、テトラヒドロピラニル基、2,3-ジヒドロベンゾフラニル基、チオフェニル基、ジイソプロピルアミノ基、メチル(フェニル)アミノ基、ピぺラジニル基、2-オキソ-1-ピロリジニル基、モルホリニル基、1,1-ジオキソチオモルホリニル基、イミダゾリル基、ピリジル基、2-メトキシエチレンオキシ基、ジメチルアミノエチレンオキシ基、ジエチルアミノエチレンオキシ基、1,2,3,4-テトラヒドロイソキノリニル基、アゼパニル基、ピラゾリル基、2-オキソイミダゾリジニル基、ジエチルアミノ基、メトキシ基、またはN-メチルピラゾリル基で示される前記〔3〕記載の化合物、
〔5〕 R2が、1~6個のR12で置換されているC1~6のアルキル基、1~6個のR12で置換されていてもよいC2~6のアルケニル基、または1~6個のR12で置換されていてもよいC2~6のアルキニル基を表し、R12が前記〔4〕記載のR12と同じ意味である前記〔3〕記載の化合物、
〔6〕 R7が、水素原子で示される前記〔4〕記載の化合物、
〔7〕 一般式(I)で示される化合物が、
(1) 2-{3-[(4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(2) 2-{3-[(1,4-ジメチル-1H-インドール-3-イル)メチル]-6-メチルイミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(3) 2-{3-[(1,4-ジメチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(4) 2-{3-[(1-エチル-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(5) 2-{6-フルオロ-3-[(4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(6) 2-{3-[(5-フルオロ-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(7) 2-[3-({4-メチル-1-[2-(4-モルホリニル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(8) 2-(3-{[4-メチル-1-(2-プロパニル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(9) 2-{3-[(4-メチル-1-プロピル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(10) 2-{3-[(1-エチル-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(11) 2-{3-[(5-フルオロ-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(12) 2-(3-{[1-(2-ヒドロキシエチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(13) 2-[3-({1-[3-(ジメチルアミノ)-2-ヒドロキシプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(14) 2-(3-{[1-(2,3-ジヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(15) 2-[3-({1-[2-(ジメチルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(16) 2-[3-({1-[3-(ジメチルアミノ)プロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(17) 2-[3-({4-メチル-1-[2-(ピロリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(18) 2-[3-({4-メチル-1-[2-(1-メチルピロリジン-2-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(19) 2-[3-({4-メチル-1-[2-(ピぺリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(20) 2-(3-{[4-メチル-1-(2-フェノキシエチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(21) 2-[3-({1-[(3R)-3-ヒドロキシ-3-フェニルプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(22) 2-[3-({1-[(3S)-3-ヒドロキシ-3-フェニルプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(23) 2-[3-({1-[2-(4-フルオロフェニル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(24) 2-[3-({4-メチル-1-[2-(テトラヒドロ-2H-ピラン-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(25) 2-[3-({1-[2-(2,3-ジヒドロ-1-ベンゾフラン-5-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(26) 2-(3-{[4-メチル-1-(3-フェニルプロピル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(27) 2-(3-{[4-メチル-1-(ピぺリジン-4-イルメチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(28) 2-(3-{[1-(3-ヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(29) 2-[3-({1-[2-(ジプロパン-2-イルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(30) 2-[3-({4-メチル-1-[2-(ピペラジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(31) 2-[3-({4-メチル-1-[2-(2-オキソピロリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(32) 2-[3-({1-[2-(1,1-ジオキシドチオモルホリン-4-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(33) 2-[3-({1-[2-(3,4-ジヒドロイソキノリン-2(1H)-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(34) 2-{3-[(1-{2-[2-(ジエチルアミノ)エトキシ]エチル}-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(35) 2-[3-({4-メチル-1-[2-(1-メチル-1H-ピラゾール-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(36) 2-{3-[(1-{2-[2-(ジメチルアミノ)エトキシ]エチル}-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(37) 2-(3-{[4-メチル-1-(4-メチルペンタ-3-エン-1-イル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(38) 2-[3-({1-[2-(3-フルオロフェニル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(39) 2-[3-({4-メチル-1-[2-(1H-ピラゾール-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(40) 2-[3-({1-[2-(アゼパン-1-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(41) 2-{3-[(4-メチル-1-{2-[メチル(フェニル)アミノ]エチル}-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(42) 2-[3-({1-[2-(1H-イミダゾール-1-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(43) 2-[3-({4-メチル-1-[2-(ピリジン-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(44) 2-[3-({1-[2-(2-メトキシエトキシ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(45) 2-[3-({4-メチル-1-[2-(チオフェン-3-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(46) 2-[3-({4-メチル-1-[2-(チオフェン-2-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(47) 2-(3-{[4-メチル-1-(3-メチルブタ-3-エン-1-イル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(48) 2-[3-({4-メチル-1-[2-(2-オキソイミダゾリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(49) 2-(3-{[1-(ブタ-3-エン-1-イル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(50) 2-[3-({4-メチル-1-[(3Z)-ペンタ-3-エン-1-イル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(51) 2-[3-({4-メチル-1-[3-(ピリジン-2-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(52) 2-(3-{[1-(3,4-ジヒドロキシブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(53) 2-[3-({1-[2-(ジエチルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(54) 2-(3-{[1-(4-フルオロブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(55) 2-(3-{[4-メチル-1-(4,4,4-トリフルオロブチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(56) 2-(3-{[1-(4-ヒドロキシブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(57) 2-(3-{[1-(3-フルオロプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(58) 2-(3-{[4-メチル-1-(テトラヒドロ-2H-ピラン-2-イルメチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(59) 2-[3-({4-メチル-1-[3-(ピリジン-3-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(60) 2-[3-({4-メチル-1-[3-(ピリジン-4-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(61) 2-[3-({1-[3-(1H-イミダゾール-1-イル)プロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(62) 2-(3-{[1-(2-ヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(63) 2-(3-{[1-(2-ヒドロキシ-3-メトキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(64) 2-(3-{[1-(2-ヒドロキシ-2-メチルプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、または
(65) 2-(3-{[1-(2-ヒドロキシ-3-フェノキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸である前記〔6〕記載の化合物、
〔8〕 前記〔1〕記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグを有効成分として含有してなる医薬組成物、
〔9〕 キマーゼ阻害剤である前記〔8〕記載の医薬組成物、
〔10〕 キマーゼ介在性疾患の予防および/または治療剤である前記〔8〕記載の医薬組成物、
〔11〕 キマーゼ介在性疾患がアトピー性皮膚炎、潰瘍性大腸炎、心不全、肺線維症、動脈瘤、非アルコール性脂肪性肝炎、消化性潰瘍、またはアレルギー性結膜炎である前記〔10〕記載の医薬組成物、
〔12〕 前記〔1〕記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグと、非ステロイド系抗炎症薬、ステロイド薬、免疫抑制薬、プロスタグランジン類、抗アレルギー薬、メディエーター遊離抑制薬、ロイコトリエン受容体拮抗薬、抗ヒスタミン薬、オピオイド作用薬、ホスホジエステラーゼ阻害薬、フォルスコリン製剤、一酸化窒素合成酵素阻害薬、カンナビノイド-2受容体刺激薬、デコイ製剤、アミノサリチル酸製剤、利尿薬、アンジオテンシン変換酵素阻害薬、アンジオテンシンII受容体拮抗薬、抗不整脈薬、ジギタリス製剤、キマーゼ阻害薬、レニン阻害薬、カルシウム拮抗薬、ベータ遮断薬、硝酸薬、抗アルドステロン薬、強心薬、抗血小板薬、抗凝固薬、抗線維化薬、血糖降下薬、血圧低下薬、脂質改善薬、抗肥満薬、肝庇護薬、および抗酸化薬から選ばれる少なくとも1種以上と組み合わせてなる医薬、
〔13〕 前記〔1〕記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグの有効量を患者に投与することを特徴とするキマーゼ介在性疾患の予防および/または治療方法、
〔14〕 キマーゼ介在性疾患を予防および/または治療するための、前記〔1〕記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグ、
〔15〕 キマーゼ介在性疾患の予防および/または治療剤の製造のための前記〔1〕記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグの使用に関する。
より好ましくは、
メトキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、2-(3-{[1-(2-ヒドロキシ-2-メチルプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、または2-(3-{[1-(2-ヒドロキシ-3-フェノキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグである。
一般式(I)で示される化合物は、公知の方法で塩に変換される。
一般式(I)で示される化合物のプロドラッグとは、生体内において酵素や胃酸等による反応により一般式(I)で示される化合物に変換される化合物をいう。一般式(I)で示される化合物のプロドラッグとしては、一般式(I)で示されるカルボキシ基が、エステル化、またはアミド化された化合物(例えば、一般式(I)で示される化合物のカルボキシ基がエチルエステル化、イソプロピルエステル化、フェニルエステル化、カルボキシメチルエステル化、ジメチルアミノメチルエステル化、ピバロイルオキシメチルエステル化、エトキシカルボニルオキシエチルエステル化、フタリジルエステル化、(5-メチル-2-オキソ-1,3-ジオキソレン-4-イル)メチルエステル化、シクロヘキシルオキシカルボニルエチルエステル化、メチルアミド化された化合物など);一般式(I)で示されるカルボキシル基が、ヒドロキシメチル基と置き換わった化合物等が挙げられる。これらの化合物はそれ自体公知の方法によって製造することができる。また、一般式(I)で示される化合物のプロドラッグは水和物および非水和物のいずれであってもよい。
一般式(I)で示される本発明化合物は、公知の方法、例えば以下に示す方法、これらに準ずる方法または実施例に示す方法に従って製造することができる。なお、以下の各製造方法において、原料化合物は塩として用いてよい。また、原料化合物が反応性官能基(カルボキシル基等)を有する場合には、必要に応じ該反応性官能基を適切な保護基で保護し、反応終了後に該保護基を除去することもできる。なお、原料化合物が塩として用いられる場合、該塩としては、一般式(I)の薬学的に許容される塩として記載されたものが用いられる。
(1)アルカリ加水分解による脱保護反応、
(2)酸性条件下における脱保護反応、
(3)加水素分解による脱保護反応、
(4)脱シリル化反応、
(5)金属を用いた脱保護反応、
(6)金属錯体を用いた脱保護反応等が挙げられる。
(1)アルカリ加水分解による脱保護反応は、例えば、有機溶媒(メタノール、テトラヒドロフラン、ジオキサン等)中、アルカリ金属の水酸化物(水酸化ナトリウム、水酸化カリウム、水酸化リチウム等)、アルカリ土類金属の水酸化物(水酸化バリウム、水酸化カルシウム等)または炭酸塩(炭酸ナトリウム、炭酸カリウム等)あるいはその水溶液もしくはこれらの混合物を用いて、約0~150℃の温度で行なわれる。
本発明化合物の毒性は十分に低いものであり、医薬品として使用するために十分安全であることが確認された。
本発明化合物は、キマーゼ阻害活性を示すため、キマーゼ阻害介在性疾患、例えば、皮膚疾患、循環器系疾患、消化器系疾患、呼吸器系疾患、腎疾患、眼疾患、肝疾患、外科手術に起因する臓器癒着等の予防および/または治療剤として有用である。
1)その化合物の予防および/または治療効果の補完および/または増強、
2)その化合物の動態・吸収改善、投与量の低減、および/または
3)その化合物の副作用の軽減のために他の薬剤と組み合わせて、併用剤として投与してもよい。
アルゴン雰囲気下、メチル 2-トリフルオロメタンスルホニルオキシ-1-ナフトエート(3.34g)のアセトニトリル(20mL)溶液に、ブチルビニルエーテル(6.4mL)、酢酸パラジウム(0.17g)、トリフェニルホスフィン(0.39g)、トリエチルアミン(1.7mL)を加えて、還流下で終夜撹拌した。反応混合物をセライト(登録商標)にてろ過し、ろ液を減圧下濃縮した。得られた残渣をテトラヒドロフラン(200mL)に溶解し、氷冷した。反応混合物に1M塩酸(200mL)を加えて、10分間撹拌した。反応混合物に水を加えて、酢酸エチルで抽出した。得られた有機層を水および飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥後、濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=75:25)で単離精製することにより、以下の物性値を有する標題化合物(2.15g)を得た。
TLC:Rf 0.36(ヘキサン:酢酸エチル=3:1);
1H-NMR(CDCl3):δ 2.70 (s, 3 H), 4.07 (s, 3 H), 7.52-7.68 (m, 2 H), 7.85-8.06 (m, 4 H)。
実施例1で製造された化合物(454mg)の1,2-ジメトキシエタン(10mL)溶液にジイソプロピルエチルアミン(0.10mL)、フェニルトリメチルアンモニウムトリブロミド(747mg)を加え、室温で3.5時間撹拌した。反応混合物をろ過し、ろ液を減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=75:25)で単離精製することにより、以下の物性値を有する標題化合物(325mg)を得た。
TLC:Rf 0. 40(ヘキサン:酢酸エチル=3:1);
1H-NMR(CDCl3):δ 4.07 (s, 3 H), 4.51 (s, 2 H), 7.57-7.70 (m, 2 H), 7.81 (d, 1 H), 7.87-7.95 (m, 1 H), 7.97-8.10 (m, 2 H)。
実施例2で製造された化合物(321mg)のジメチルホルムアミド(10mL)溶液に、2-アミノピリジン(184mg)を加え、80℃で1時間撹拌した。反応混合物に水を加えて酢酸エチルで抽出した。得られた有機層を飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=50:50)で単離精製することにより、以下の物性値を有する標題化合物(145mg)を得た。
TLC:Rf 0.37(ヘキサン:酢酸エチル=1:1);
1H-NMR(CDCl3):δ 4.01 (s, 3 H), 6.73-6.87 (m, 1 H), 7.10-7.23 (m, 1 H), 7.44-7.60 (m, 2 H), 7.64 (dd, 1 H), 7.79-7.91 (m, 3 H), 7.91-8.04 (m, 2 H), 8.08-8.22 (m, 1 H)。
実施例3で製造された化合物(172mg)をオキシ塩化リン(1.7mL)に懸濁し、室温で撹拌した。ジメチルホルムアミド(0.046mL)を加えて70℃で5.5時間撹拌した。反応混合物を氷冷し、2M水酸化ナトリウム水溶液で中和後、酢酸エチルで抽出した。得られた有機層を飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥後、減圧下濃縮することにより、以下の物性値を有する標題化合物(196mg)を得た。
TLC:Rf 0.37(ヘキサン:酢酸エチル=1:1);
1H-NMR(CDCl3):δ 3.82 (s, 3 H), 7.11-7.22 (m, 1 H), 7.52-7.68(m, 3 H), 7.72 (d, 1 H), 7.78-7.87 (m, 1 H), 7.91-8.00 (m, 1 H), 8.01-8.13 (m, 2 H), 9.58-9.71 (m, 1 H), 9.95 (s, 1 H)。
実施例4で製造された化合物(195mg)をテトラヒドロフラン(1.5mL)に懸濁し、氷冷下で水素化ホウ素ナトリウム(34mg)の水(0.4mL)溶液を加えて、1時間撹拌した。反応混合物に飽和塩化アンモニウム水溶液を加え、酢酸エチルで2回抽出した。得られた有機層を合わせて、水、飽和炭酸水素ナトリウム水溶液および飽和食塩水で順次洗浄し、無水硫酸ナトリウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=25:75)で単離精製することにより、以下の物性値を有する標題化合物(186mg)を得た。
TLC:Rf 0.27(ヘキサン:酢酸エチル=1:3);
1H-NMR(CDCl3):δ 2.65 (t, 1 H), 3.77 (s, 3 H), 4.86 (d, 2 H), 6.81-6.97 (m, 1 H), 7.24-7.33 (m, 1 H), 7.51-7.69 (m, 4 H), 7.82-7.95 (m, 2 H), 7.97 (d, 1 H), 8.25-8.38 (m, 1 H)。
実施例5で製造された化合物(47mg)および4-メチルインドール(20mg)を水(0.25mL)に懸濁し、室温で撹拌した。酢酸(0.024mL)を加えて95℃で終夜撹拌した。反応混合物に飽和炭酸水素ナトリウム水溶液を加え、酢酸エチルで抽出した。得られた有機層を飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=25:75)で単離精製することにより、以下の物性値を有する標題化合物(36mg)を得た。
TLC:Rf 0.46(ヘキサン:酢酸エチル=1:3);
1H-NMR(CDCl3):δ 2.82 (s, 3 H), 3.91 (s, 3 H), 4.77 (d, 1 H), 4.81 (d, 1 H), 6.34-6.50 (m, 1 H), 6.65-6.78 (m, 1 H), 6.87-7.00 (m, 1 H), 7.06-7.25 (m, 3 H), 7.46-7.60 (m, 2 H), 7.61-7.70 (m, 2 H), 7.79-7.89 (m, 3 H), 7.92-8.06 (m, 2 H)。
TLC:Rf 0.32(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 2.68 (s, 3 H), 4.73 (s, 2 H), 6.46 (s, 1 H), 6.71 (d, 1 H), 6.93 (t, 1 H), 7.03-7.28 (m, 2 H), 7.49-7.73 (m, 4 H), 7.74-7.86 (m, 1 H), 7.93-8.15 (m, 3 H), 8.24-8.42 (m, 1 H), 10.8 (s, 1 H)。
氷冷下、2-アセチル安息香酸(70.29g)のジメチルホルムアミド(500mL)溶液に、ヨウ化メチル(66.85g)および炭酸カリウム(118.40g)を加え、1時間撹拌した。反応混合物をセライト(登録商標)にてろ過した後、ろ液を減圧下濃縮した。得られた残渣に飽和塩化アンモニウム水溶液を加えて酢酸エチルで抽出した。得られた有機層を水および飽和食塩水で洗浄して、無水硫酸ナトリウムで乾燥後、減圧下濃縮することにより、以下の物性値を有する標題化合物(72.24g)を得た。
TLC:Rf 0.40(ヘキサン:酢酸エチル=3:1);
1H-NMR(CDCl3):δ 2.54 (s, 3 H), 3.90 (s, 3 H), 7.41 (dd, 1 H), 7.45-7.61 (m, 2 H), 7.84 (dd, 1 H)。
2-アミノピリジンの代わりに5-メチル-2-アミノピリジンを用い、実施例1で製造した化合物の代わりに実施例8で製造した化合物を用いて、実施例2→実施例3→実施例4→実施例5と同様の操作を行って、以下の物性値を有する標題化合物を得た。
TLC:Rf 0.13(ヘキサン:酢酸エチル=2:3);
1H-NMR(CDCl3):δ 2.38 (s, 3 H), 2.72 (t, 1 H), 3.77 (s, 3 H), 4.77 (d, 2 H), 7.08-7.12 (m, 1 H), 7.37-7.61 (m, 4 H), 7.93-7.96 (m, 1 H), 8.08 (s, 1 H)。
4-メチルインドール(22.08g)のジメチルホルムアミド(200mL)溶液に、ヨウ化メチル(28.44g)および炭酸セシウム(82.26g)を加え、室温で終夜撹拌した。反応混合物をセライト(登録商標)にてろ過した後、ろ液を減圧下濃縮した。得られた残渣に飽和塩化アンモニウム水溶液を加えて酢酸エチルで抽出した。得られた有機層を水および飽和食塩水で洗浄して、無水硫酸ナトリウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=100:0→85:15)で単離精製することにより、以下の物性値を有する標題化合物(20.83g)を得た。
TLC:Rf 0.79(ヘキサン:酢酸エチル=4:1);
1H-NMR(CDCl3):δ 2.55 (s, 3 H), 3.78 (s, 3 H), 6.49 (dd, 1 H), 6.86-6.94 (m, 1 H), 7.04 (d, 1 H), 7.09-7.20 (m, 2 H)。
4-メチルインドールの代わりに、実施例10で製造した化合物を用い、実施例5で製造した化合物の代わりに、実施例9で製造した化合物を用いて、実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.22(塩化メチレン:メタノール=10:1);
1H-NMR(DMSO-D6):δ 2.21 (s, 3 H), 2.67 (s, 3 H), 3.50 (s, 3 H), 4.57 (s, 2 H), 6.39 (s, 1 H), 6.73 (d, 1 H), 6.89-7.05 (m, 2 H), 7.11 (d, 1 H), 7.18-7.35 (m, 3 H), 7.42-7.61 (m, 2 H), 7.89 (s, 1 H)。
実施例1で製造した化合物の代わりに、実施例8で製造した化合物を用いて、実施例2→実施例3→実施例4→実施例5と同様の操作を行って、以下の物性値を有する標題化合物を得た。
TLC:Rf 0.45(ヘキサン:酢酸エチル=1:4)。
1H-NMR(CDCl3):δ 2.78 (s, 3 H), 3.57 (s, 3 H), 3.71 (s, 3 H), 4.67 (d, 2 H), 6.26 (s, 1 H), 6.65-6.76 (m, 1 H), 6.82-6.92 (m, 1 H), 7.07-7.22 (m, 3 H), 7.34-7.47 (m, 2 H), 7.47-7.54 (m, 1 H), 7.59-7.69 (m, 1 H), 7.77-7.88 (m, 2 H)。
実施例5で製造した化合物の代わりに、実施例12で製造した化合物を用い、4-メチルインドールの代わりに、実施例10で製造した化合物を用いて、実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.27(酢酸エチル:メタノール=4:1);
1H-NMR(CDCl3):δ 2.79 (s, 3 H), 3.62 (s, 3 H), 4.62-4.85 (m, 2 H), 6.26 (s, 1 H), 6.86-6.97 (m, 2 H), 7.16-7.22 (m, 2 H), 7.34-7.56 (m, 4 H), 7.69-7.79 (m, 1 H), 7.89-8.00 (m, 1 H), 8.28-8.37 (m, 1 H)。
ヨウ化メチルの代わりに、ヨウ化エチルを用いて、実施例10と同様の操作を行って、以下の物性値を有する標題化合物を得た。
TLC:Rf 0.48(ヘキサン:酢酸エチル=29:1);
1H-NMR(CDCl3):δ 1.46 (t, 3 H), 2.55 (s, 3 H), 4.17 (q, 2 H), 6.48-6.52 (m, 1 H), 6.86-6.93 (m, 1 H), 7.07-7.16 (m, 2 H), 7.20 (d, 1 H)。
4-メチルインドールの代わりに、実施例14で製造した化合物を用い、実施例5で製造した化合物の代わりに、実施例12で製造した化合物を用いて、実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.31(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 1.10 (t, 3 H), 2.67 (s, 3 H), 3.92 (q, 2 H), 4.65 (s, 2 H), 6.46 (s, 1 H), 6.73 (d, 1 H), 6.97 (t, 2 H), 7.17 (d, 1 H), 7.31-7.56 (m, 4 H), 7.67 (d, 1 H), 7.77 (d, 1 H), 8.24 (d, 1 H)。
2-アミノピリジンの代わりに、5-フルオロ-2-アミノピリジンを用いて、実施例3→実施例4→実施例5→実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.43(塩化メチレン:メタノール=10:1);
1H-NMR(DMSO-D6):δ 2.69 (s, 3 H), 4.74 (s, 2 H), 6.35 (s, 1 H), 6.71 (d, 1 H), 6.93 (t, 1 H), 7.11 (d, 1 H), 7.35-7.49 (m, 1 H), 7.53-7.67 (m, 3 H), 7.69-7.81 (m, 1 H), 7.91-8.06 (m, 3 H), 8.41-8.50 (m, 1 H), 10.7 (d, 1 H)。
アルゴン雰囲気下、4-クロロ-2-フルオロ-5-ニトロトルエン(1.9g)を無水テトラヒドロフラン(100mL)に溶解し、-50℃で撹拌した。反応混合物にビニルマグネシウムブロマイド(1.0Mテトラヒドロフラン溶液)40mLを滴下した。反応混合物を40分かけて-20℃まで徐々に昇温させた。反応混合物に飽和塩化アンモニウム水溶液を加え、酢酸エチルで抽出した。得られた有機層を水および飽和食塩水で洗浄して、硫酸マグネシウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=95:5→91:9)で単離精製することにより、以下の物性値を有する標題化合物(1.09g)を得た。
TLC:Rf 0.50(ヘキサン:酢酸エチル=5:1);
1H-NMR(CDCl3):δ 2.42 (d, 3 H), 6.58 (dd, 1 H), 6.98 (d, 1 H), 7.26-7.31 (m, 1 H), 8.30 (br s, 1 H)。
実施例17で製造された化合物(500mg)をエタノール(9mL)に溶解し、トリエチルアミン(0.46mL)を加えて室温で撹拌した。アルゴン雰囲気下、10%Pd-C(50%wet)を50mg加えた。反応混合物をアルゴン雰囲気から水素雰囲気にした後、4.5時間撹拌した。反応混合物をセライト(登録商標)にてろ過し、ろ液を減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=98:2→91:9)で単離精製することにより、以下の物性値を有する標題化合物(355mg)を得た。
TLC:Rf 0.37(ヘキサン:酢酸エチル=5:1);
1H-NMR(CDCl3):δ 2.46 (d, 3 H), 6.48-6.59 (m, 1 H), 6.86-6.97 (m, 1 H), 7.12-7.18 (m, 1 H), 7.22-7.25 (m, 1 H), 8.12 (br s, 1 H)。
4-メチルインドールの代わりに、実施例18で製造した化合物を用い、実施例5で製造した化合物の代わりに、実施例12で製造した化合物を用いて、実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.56(塩化メチレン:メタノール=10:1);
1H-NMR(CDCl3):δ 2.66 (s, 3 H), 4.68 (s, 2 H), 6.44 (s, 1 H), 6.87 (t, 1 H), 6.97 (t, 1 H), 7.13 (dd, 1 H), 7.29-7.38 (m, 1 H), 7.39-7.52 (m, 3 H), 7.73 (d, 1 H), 7.89 (d, 1 H), 8.12 (br s, 1 H), 8.26 (d, 1 H)。
実施例7で製造された化合物(190mg)を4N塩酸/1,4-ジオキサン溶液(9mL)に溶解させ、室温で5分撹拌した。その後、反応混合物を減圧下濃縮した。得られた残渣をメタノールに溶解させ、再度、減圧下濃縮することにより、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.38(塩化メチレン:メタノール=6:1);
1H-NMR(DMSO-D6):δ 2.65 (s, 3 H), 4.72 (s, 2 H), 6.59 (s, 1 H), 6.70 (d, 1 H), 6.92 (dd, 1 H), 7.11 (d, 1 H), 7.29-7.49 (m, 1 H), 7.59-7.78 (m, 3 H), 7.78-8.23 (m, 5 H), 8.44-8.67 (m, 1 H), 10.8 (d, 1 H)。
実施例7で製造された化合物(5.0g)のエタノール(100mL)/水(50mL)溶液に、1N水酸化ナトリウム水溶液(11.6mL)を加え、80℃で還流した。反応混合物を室温まで放冷した後、減圧下濃縮した。得られた残渣に水(30mL)およびエタノール(10mL)を加え、還流した。反応混合物を室温まで放冷することにより、析出した結晶をろ過した。得られた結晶を100℃で減圧下乾燥し、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.50(塩化メチレン:メタノール:酢酸=9:1:0.1);
1H-NMR(DMSO-D6):δ 2.66 (s, 3 H), 4.75 (s, 2 H), 6.24 (d, 1 H), 6.66 (d, 1 H), 6.73-6.81 (m, 1 H), 6.87 (dd, 1 H), 7.06 (d, 1 H), 7.16 (dd, 1 H), 7.33-7.50 (m, 3 H), 7.55-7.69 (m, 2 H), 7.73-7.83 (m, 1 H), 7.99-8.17 (m, 2 H), 10.6 (s, 1 H)。
実施例13で製造した化合物を用いて、実施例21と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.57(塩化メチレン:メタノール:酢酸=9:1:0.1);
1H-NMR(DMSO-D6):δ 2.65 (s, 3 H), 3.49 (s, 3 H), 4.58 (s, 2 H), 6.48 (s, 1 H), 6.66-6.83 (m, 2 H), 6.95 (dd, 1 H), 7.05-7.23 (m, 4 H), 7.28 (dd, 1 H), 7.45 (d, 1 H), 7.53 (d, 1 H), 8.00 (d, 1 H)。
ヨウ化メチルの代わりに、相当するアルキルハライドを用いて、実施例10→実施例7→実施例20と同様の操作を行って、以下の物性値を有する本発明化合物を得た(但し、実施例23-(4)は、実施例10→実施例7と同様の操作を行って得た)。
TLC:Rf 0.35(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 1.10 (d, 6 H), 2.68 (s, 3 H), 4.31-4.60 (m, 1 H), 4.76 (s, 2 H), 6.48 (s, 1 H), 6.72 (d, 1 H), 6.84-7.02 (m, 2 H), 7.18 (d, 1 H), 7.24-7.41 (m, 1 H), 7.48-7.73 (m, 4 H), 7.86-8.12 (m, 3 H), 8.25 (d, 1 H)。
TLC:Rf 0.40(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 0.58 (t, 3 H), 1.34-1.58 (m, 2 H), 2.69 (s, 3 H), 3.85 (t, 2 H), 4.74 (s, 2 H), 6.39 (s, 1 H), 6.74 (d, 1 H), 6.89 (t, 1 H), 6.93-7.04 (m, 1 H), 7.18 (d, 1 H), 7.22-7.35 (m, 1 H), 7.49-7.72 (m, 4 H), 7.87-8.09 (m, 3 H), 8.19 (d, 1 H)。
TLC:Rf 0.51(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 1.06 (t, 3 H), 2.62 (s, 3 H), 3.88 (q, 2 H), 4.71 (s, 2 H), 6.65 (s, 1 H), 6.72 (d, 1 H), 6.91-7.02 (m, 1 H), 7.15 (d, 1 H), 7.34-7.47 (m, 1 H), 7.59 (d, 1 H), 7.62-7.75 (m, 2 H), 7.81-7.95 (m, 1 H), 7.96-8.16 (m, 4 H), 8.61 (d, 1 H)。
TLC:Rf 0.52(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 2.12-2.27 (m, 4 H), 2.40 (t, 2 H), 2.70 (s, 3 H), 3.20-3.35 (m, 4 H), 4.01 (t, 2 H), 4.73 (s, 2 H), 6.43 (s, 1 H), 6.74 (d, 1 H), 6.88 (t, 1 H), 6.94-7.03 (m, 1 H), 7.20 (d, 1 H), 7.23-7.36 (m, 1 H), 7.47-7.73 (m, 4 H), 7.88-7.98 (m, 2 H), 8.01 (d, 1 H), 8.18 (d, 1 H)。
4-メチルインドールの代わりに、実施例18で製造した化合物を用いて、実施例6→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.69(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 2.56 (s, 3 H), 4.74 (s, 2 H), 6.34 (s, 1 H), 6.77-6.91 (m, 2 H), 7.02-7.13 (m, 1 H), 7.18-7.30 (m, 1 H), 7.45-7.66 (m, 4 H), 7.79-7.94 (m, 2 H), 8.04 (d, 1 H), 8.12 (d, 1 H), 10.8 (s, 1 H)。
ヨウ化メチルの代わりに、2-(2-ブロモエトキシ)テトラヒドロ-2H-ピランを用いて、実施例10と同様の操作を行って、以下の物性値を有する標題化合物を得た。
TLC:Rf 0.33(ヘキサン:酢酸エチル=1:1);
1H-NMR(CDCl3):δ 1.17-1.63 (m, 6 H), 2.80 (s, 3 H), 3.27-3.38 (m, 1 H), 3.48-3.63 (m, 2 H), 3.83-3.94 (m, 4 H), 4.02-4.21 (m, 2 H), 4.32-4.40 (m, 1 H), 4.79 (s, 2 H), 6.43 (s, 1 H), 6.72 (td, 1 H), 6.86-6.94 (m, 1 H), 7.09-7.24 (m, 3 H), 7.46-7.60 (m, 2 H), 7.62-7.71 (m, 2 H), 7.78-7.91 (m, 3 H), 7.96-8.05 (m, 1 H)。
実施例25で製造した化合物(390mg)を酢酸12mLと水3mLに溶解させ、80℃で1.5時間撹拌した。反応混合物を室温へ冷却後、2N水酸化ナトリウム水溶液を加えて中和し、酢酸エチルで抽出した。得られた有機層を飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=80:20→50:50→0:100、酢酸エチル:メタノール=95:5→90:10)で単離精製することにより、以下の物性値を有する標題化合物(320mg)を得た。
TLC:Rf 0.22(ヘキサン:酢酸エチル=1:4);
1H-NMR(CDCl3):δ 3.49 (s, 3 H), 3.75-3.83 (m, 2 H), 3.88 (s, 3 H), 4.01-4.09 (m, 2 H), 4.74-4.81 (m, 2 H), 6.36 (s, 1 H), 6.74 (td, 1 H), 6.86-6.93 (m, 1 H), 7.08-7.24 (m, 3 H), 7.45-7.59 (m, 2 H), 7.62 (d, 1 H), 7.68 (dt, 1 H), 7.79-7.91 (m, 3 H), 7.94-8.02 (m, 1 H)。
実施例26で製造した化合物を用いて、実施例7→実施例20と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.23(クロロホルム:メタノール=9:1);
1H-NMR(DMSO-D6):δ 2.62 (s, 3 H), 3.47 (t, 2 H), 3.90 (t, 2 H), 4.70 (s, 2 H), 6.66 (s, 1 H), 6.71 (d, 1 H), 6.90-7.01 (m, 1 H), 7.17 (d, 1 H), 7.36-7.49 (m, 1 H), 7.61 (d, 1 H), 7.64-7.78 (m, 2 H), 7.87-8.17 (m, 5 H), 8.63 (d, 1 H)。
氷冷下、実施例6で製造した化合物(150mg)のジメチルホルムアミド(3.4mL)溶液に、60%濃度の水素化ナトリウム(16mg)を加え、数分間撹拌した。氷冷下、反応混合物にエピブロモヒドリン(35μL)を加え30分間撹拌した。反応混合物に1N水酸化ナトリウム水溶液(0.5mL)を加え、70℃で終夜撹拌した。反応混合物に1N塩酸を加えて、反応混合物を酸性とした後、水層を塩化メチレンで洗浄した。次いで水層を1N水酸化ナトリウム水溶液にて塩基性とした後、塩化メチレンで抽出した。得られた有機層を無水硫酸ナトリウムで乾燥した後、減圧下濃縮することで、以下の物性値を有する標題化合物(130mg)を得た。
TLC:Rf 0.45(塩化メチレン:メタノール=9:1);
1H-NMR(CDCl3):δ 2.01 (dd, 1H), 2.08-2.16 (m, 7H), 2.80 (s, 3H), 3.25 (br, 1H), 3.78-3.88 (m, 1H), 3.91 (s, 3H), 3.92-4.00 (m, 2H), 4.79 (s, 2H), 6.35 (s, 1H), 6.72 (t, 1H), 6.90 (d, 1H), 7.09-7.25 (m, 3H), 7.45-7.59 (m, 2H), 7.60-7.73 (m, 2H), 7.77-7.91 (m, 3H), 8.00 (d, 1H)。
実施例28で製造した化合物を用いて、実施例7→実施例21と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.32(酢酸エチル:酢酸:水=3:1:1);
1H-NMR(DMSO-D6):δ 1.93-2.06 (m, 8 H), 2.66 (s, 3 H), 3.60-3.81 (m, 2 H), 3.93-4.07 (m, 1 H), 4.69 (s, 2 H), 5.16-5.28 (m, 1 H), 6.38 (s, 1 H), 6.68 (d, 1 H), 6.78 (t, 1 H), 6.87-6.97 (m, 1 H), 7.08-7.20 (m, 2 H), 7.35-7.47 (m, 3 H), 7.57-7.66 (m, 2 H), 7.72-7.83 (m, 1 H), 8.02-8.18 (m, 2 H)。
実施例6で製造した化合物(150mg)のジメチルアセトアミド(5.0mL)溶液に、60%濃度の水素化ナトリウム(16mg)を室温で加え、数分間撹拌した。次いで、反応混合物にグリシドール(0.1mL)を加え、80℃で30分間撹拌した。反応混合物に水を加え、酢酸エチルで抽出した。得られた有機層を飽和食塩水で洗浄し、無水硫酸ナトリウムで乾燥した後、減圧下濃縮した。得られた残渣をシリカゲルカラムクロマトグラフィー(カラム:メインカラムL、インジェクトカラムS(山善社製)、酢酸エチル:メタノール=100:0→90:10)で精製することにより、以下の物性値を有する標題化合物(68mg)を得た。
TLC:Rf 0.45(酢酸エチル);
1H-NMR(CDCl3):δ 2.80 (s, 3 H), 3.29 (dd, 1H), 3.46 (dd, 1H), 3.80-3.93 (m, 4 H), 3.93-4.03 (m, 2 H), 4.78 (s, 2H), 6.32 (s, 1H), 6.75 (t, 1H), 6.89 (d, 1H), 7.06-7.24 (m, 3H), 7.45-7.59 (m, 2H), 7.60-7.73 (m, 2H), 7.80-7.91 (m, 3H), 7.96 (d, 1H)。
実施例30で製造した化合物を用いて、実施例7→実施例20と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.36(塩化メチレン:メタノール=9:1);
1H-NMR(DMSO-D6):δ 2.63 (s, 3 H), 3.07-3.27 (m, 2 H), 3.51-3.63 (m, 1 H), 3.74 (dd, 1 H), 4.00 (dd, 1 H), 4.70 (s, 2 H), 6.57 (s, 1 H), 6.72 (d, 1 H), 6.90-7.05 (m, 1 H), 7.19 (d, 1 H), 7.21-7.38 (m, 1 H), 7.55-8.19 (m, 8 H), 8.39-8.56 (m, 1 H)。
ヨウ化メチルの代わりに、相当するアルキルハライドを用いて、実施例10→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.39(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 2.39 (s, 6H), 2.67 (s, 3H), 2.95-3.00 (m, 2H), 4.10-4.15 (m, 2H), 4.46 (s, 2H), 6.66 (s, 1H), 6.72-6.75 (m, 1H), 6.79-6.81 (m, 1H), 7.05-7.06 (m, 2H), 7.18-7.22 (m, 1H), 7.42-7.48 (m, 2H), 7.58 (d, 1H), 7.70 (d, 1H), 7.83-7.89 (m, 3H), 8.11 (d, 1H)。
TLC:Rf 0.24(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 1.81-1.92 (m, 2H), 2.35 (s, 6H), 2.50-2.60 (m, 2H), 2.77 (s, 3H), 3.36-3.43 (m, 2H), 4.65 (s, 2H), 6.63-6.67 (m, 2H), 6.82 (d, 1H), 6.88 (d, 1H), 7.04 (dd, 1H), 7.15-7.22 (m, 1H), 7.38-7.46 (m, 2H), 7.54 (d, 1H), 7.67 (d, 1H), 7.74-7.82 (m, 3H), 8.13 (d, 1H)。
TLC:Rf 0.50(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 1.79-1.90 (m, 4H), 2.68 (s, 3H), 2.82-3.00 (m, 2H), 3.19-3.30 (m, 2H), 4.09-4.19 (m, 2H), 4.48 (s, 2H), 6.70-6.75 (m, 2H), 6.77-6.81 (m, 1H), 7.02-7.05 (m, 2H), 7.14-7.19 (m, 1H), 7.39-7.45 (m, 2H), 7.52 (d, 1H), 7.61 (d, 1H), 7.76 (d, 1H), 7.80 (d, 1H), 7.87 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.23(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.19-1.28 (m, 1H), 1.43-1.56 (m, 3H), 1.60-1.69 (m, 1H), 1.81-1.89 (m, 1H), 1.91-2.00 (m, 1H), 2.02-2.08 (m, 4H), 2.66 (s, 3H), 2.83-2.88 (m, 1H), 3.87-3.91 (m, 2H), 4.70 (s, 2H), 6.41 (s, 1H), 6.73 (d, 1H), 6.82 (dd, 1H), 6.97 (dd, 1H), 7.12 (d, 1H), 7.23 (dd, 1H), 7.50-7.60 (m, 4H), 7.87-7.92 (m, 2H), 8.03 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.53(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 1.54-1.66 (m, 2 H), 1.72-1.85 (m, 4 H), 2.71 (s, 3 H), 2.97-3.12 (m, 4 H), 3.37-3.46 (m, 2 H), 4.28-4.41 (m, 2 H), 4.62 (s, 2 H), 6.71 (s, 1 H), 6.78 (d, 1 H), 6.84-6.93 (m, 1 H), 6.98-7.08 (m, 1 H), 7.21 (d, 1 H), 7.30-7.40 (m, 1 H), 7.43-7.51 (m, 2 H), 7.55 (d, 1 H), 7.65 (d, 1 H), 7.86 (d, 2 H), 8.05 (d, 1 H), 8.15 (d, 1 H)。
TLC:Rf 0.70(クロロホルム:メタノール:酢酸=79:20:1);
1H-NMR(CDCl3):δ 2.65 (s, 3H), 4.12-4.18 (m, 2H), 4.33-4.41 (m, 2H), .7.23 (s, 2H), 6.12 (s, 1H), 6.63-6.71 (m, 4H), 6.85-6.95 (m, 4H), 7.13 (dd, 1H), 7.19-7.23 (m, 2H), 7.27-7.38 (m, 2H), 7.46 (d, 1H), 7.68 (d, 1H), 7.71 (d, 1H), 7.96-8.02 (m, 1H), 8.10 (d, 1H)。
TLC:Rf 0.50(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.81-1.87 (m, 2H), 2.64 (s, 3H), 3.88-4.06 (m, 2H), 4.33-4.47 (m, 1H), 4.70 (s, 2H), 6.44 (s, 1H), 6.71 (d, 1H), 6.79-6.82 (m, 1H), 6.92-6.96 (m, 1H), 7.08 (d, 1H), 7.12-7.17 (m, 3H), 7.19-7.23 (m, 3H), 7.47-7.59 (m, 4H), 7.81 (d, 1H), 7.86 (d, 1H), 8.04 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.50(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.81-1.87 (m, 2H), 2.64 (s, 3H), 3.88-4.06 (m, 2H), 4.33-4.47 (m, 1H), 4.70 (s, 2H), 6.44 (s, 1H), 6.71 (d, 1H), 6.79-6.82 (m, 1H), 6.92-6.96 (m, 1H), 7.08 (d, 1H), 7.12-7.17 (m, 3H), 7.19-7.23 (m, 3H), 7.47-7.59 (m, 4H), 7.81 (d, 1H), 7.86 (d, 1H), 8.04 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.50(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 2.64 (s, 3H), 2.81 (t, 2H), 4.10 (t, 2H), 4.67 (s, 2H), 6.33 (s, 1H), 6.71 (d, 1H), 6.81-6.87 (m, 3H), 6.92-7.04 (m, 3H), 7.12 (d, 1H), 7.22-7.26 (m, 1H), 7.50-7.59 (m, 4H), 7.87-7.92 (m, 2H), 8.03-8.05 (m, 2H)。
TLC:Rf 0.49(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 0.99-1.10 (m, 2H), 1.18-1.27 (m, 1H), 1.36-1.39 (dd, 2H), 1.43-1.48 (dt, 2H), 2.66 (s, 3H), 3.05 (t, 2H), 3.65-3.69 (m, 2H), 3.88 (t, 2H), 4.71 (s, 2H), 6.39 (s, 1H), 6.72 (d, 1H), 6.81-6.84 (m, 1H), 6.94-6.97 (m, 1H), 7.11 (d, 1H), 7.21-7.25 (m, 1H), 7.49-7.59 (m, 4H), 7.86 (d, 1H), 7.90 (d, 1H), 8.05 (d, 1H), 8.10 (d, 1H)。
TLC:Rf 0.54(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.64 (s, 3H), 2.72 (t, 2H), 2.99 (t, 2H), 4.04 (t, 2H), 4.40 (t, 2H), 4.68 (s, 2H), 6.38 (s, 1H), 6.46 (d, 1H), 6.67 (d, 1H), 6.71 (d, 1H), 6.79-6.82 (m, 1H), 6.87 (s, 1H), 6.94 (dd, 1H), 7.12 (d, 1H), 7.20-7.24 (m, 1H), 7.48-7.58 (m, 4H), 7.82 (d, 1H), 7.88 (d, 1H), 8.03-8.07 (m, 2H)。
TLC:Rf 0.53(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.80-1.87 (m, 2H), 2.35 (t, 2H), 2.65 (s, 3H), 3.87 (t, 2H), 4.72 (s, 2H), 6.40 (s, 1H), 6.72 (d, 1H), 6.79-6.83 (m, 1H), 6.93-6.97 (m, 3H), 7.07-7.12 (m, 2H), 7.15-7.24 (m, 3H), 7.48-7.60 (m, 4H), 7.83 (d, 1H), 7.87 (d, 1H), 8.04 (d, 1H), 8.12 (d, 1H)。
TLC:Rf 0.53(クロロホルム:エタノール:水=3:7:4);
1H-NMR(DMSO-D6):δ 0.99-1.11 (m, 2H), 1.27-1.31 (m, 2H), 1.70-1.80 (m, 1H), 2.40-2.50 (d, 2H), 2.66 (s, 3H), 2.91-2.94 (m, 2H), 3.75 (d, 2H), 4.69 (s, 2H), 6.34 (s, 1H), 6.72 (d, 1H), 6.79-6.82 (m, 1H), 6.93-6.97 (m, 1H), 7.13 (d, 1H), 7.20-7.24 (m, 1H), 7.49-7.59 (m, 4H), 7.81 (d, 1H), 7.86 (d, 1H), 8.07-8.08 (m, 2H)。
TLC:Rf 0.23(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 1.63-1.69 (m, 2H), 2.64 (s, 3H), 3.23 (t, 2H), 3.92 (t, 2H), 4.69 (s, 2H), 6.42 (s, 1H), 6.71 (d, 1H), 6.80-6.83 (m, 1H), 6.95 (dd, 1H), 7.13 (d, 1H), 7.19-7.23 (m, 1H), 7.49-7.59 (m, 4H), 7.84 (d, 1H), 7.88 (d, 1H), 8.05-8.09 (m, 2H)。
TLC:Rf 0.35(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 0.68 (d, 12H), 2.71 (s, 3H), 2.77-2.85 (m, 2H), 3.15-3.62 (m, 2H), 3.80-3.90 (m, 2H), 4.73 (s, 2H), 6.40 (s, 1H), 6.74 (d, 1H), 6.86-6.89 (m, 1H), 6.97-7.01 (m, 1H), 7.16 (d, 1H), 7.25-7.29 (m, 1H), 7.54-7.65 (m, 4H), 7.92-8.02 (m, 3H), 8.17 (d, 1H)。
TLC:Rf 0.44(クロロホルム:エタノール:水=3:7:4);
1H-NMR(DMSO-D6):δ 2.31-2.33 (m, 4H), 2.50 (t, 2H), 2.62-2.69 (m, 7H), 3.97 (t, 2H), 4.69 (s, 2H), 6.48 (s, 1H), 6.73 (d, 1H), 6.80-6.84 (m, 1H), 6.94-6.98 (m, 1H), 7.15 (s, 1H), 7.21-7.25 (m, 1H), 7.50-7.59 (m, 4H), 7.86-7.91 (m, 2H), 8.05-8.07 (m, 2H)。
TLC:Rf 0.44(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.48-1.55 (m, 2H), 1.91 (m, 2H), 2.66 (s, 3H), 2.80 (t, 2H), 3.30 (t, 2H), 4.01 (t, 2H), 4.71 (s, 2H), 6.40 (s, 1H), 6.75 (d, 1H), 6.82-6.85 (m, 1H), 6.96-7.00 (m, 1H), 7.15 (d, 1H), 7.22-7.26 (m, 1H), 7.50-7.61 (m, 4H), 7.88-7.92 (m, 2H), 8.03 (d, 1H), 8.11 (d, 1H)。
TLC:Rf 0.26(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.63 (t, 2H), 2.66 (s, 3H), 2.71-2.78 (m, 8H), 3.95 (t, 2H), 4.71 (s, 2H), 6.46 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.94-6.98 (m, 1H), 7.15 (d, 1H), 7.20-7.24 (m, 1H), 7.49-7.59 (m, 4H), 7.86 (d, 1H), 7.89 (d, 1H), 8.05 (d, 1H), 8.11 (d, 1H)。
TLC:Rf 0.43(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.55-2.57 (m, 4H), 2.62-2.66 (m, 5H), 3.46 (s, 2H), 4.06 (t, 2H), 4.68 (s, 2H), 6.48 (s, 1H), 6.70-6.73 (m, 2H), 6.89 (d, 1H), 6.94-6.98 (m, 2H), 7.01-7.05 (m, 2H), 7.16-7.20 (m, 2H), 7.50-7.58 (m, 4H), 7.80 (d, 1H), 7.88 (d, 1H), 8.00 (d, 1H), 8.08 (d, 1H)。
TLC:Rf 0.15(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 0.85 (t, 6H), 2.41 (q, 4H), 2.46-2.50 (m, 2H), 2.67 (s, 3H), 3.28 (t, 2H), 3.50 (t, 2H), 4.00 (t, 2H), 4.70 (s, 2H), 6.51 (s, 1H), 6.73 (d, 1H), 6.80-6.83 (m, 1H), 6.93-6.97 (m, 1H), 7.14 (d, 1H), 7.20-7.24 (m, 1H), 7.50-7.58 (m, 4H), 7.87 (d, 1H), 7.90 (d, 1H), 8.05-8.10 (m, 2H)。
TLC:Rf 0.36(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.62-2.66 (m, 5H), 3.62 (s, 3H), 4.00 (t, 2H), 4.69 (s, 2H), 6.41 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.93-6.97 (m, 1H), 7.01 (s, 1H), 7.13 (d, 1H), 7.16 (s, 1H), 7.22-7.25 (m, 1H), 7.50-7.59 (m, 4H), 7.87 (d, 1H), 7.90 (d, 1H), 8.04 (d, 1H), 8.08 (d, 1H)。
TLC:Rf 0.08(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.13 (s, 6H), 2.32 (t, 2H), 2.68 (s, 3H), 3.31 (t, 2H), 3.50 (t, 2H), 4.00 (t, 2H), 4.70 (s, 2H), 6.57 (s, 1H), 6.73 (d, 1H), 6.80-6.83 (m, 1H), 6.93-6.97 (m, 1H), 7.14 (d, 1H), 7.20-7.24 (m, 1H), 7.49-7.59 (m, 4H), 7.87-7.91 (m, 2H), 8.07-8.10 (m, 2H)。
TLC:Rf 0.69(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.30 (s, 3H), 1.45 (s, 3H), 2.19 (dt, 2H), 2.66 (s, 3H), 3.85 (t, 2H), 4.70 (s, 2H), 4.90 (t, 1H), 6.37 (s, 1H), 6.72 (d, 1H), 6.81-6.84 (m, 1H), 6.94-6.98 (m, 1H), 7.12 (d, 1H), 7.21-7.25 (m, 1H), 7.50-7.59 (m, 4H), 7.87-7.91 (m, 2H), 8.03 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.66(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 2.67 (s, 3H), 2.95 (t, 2H), 4.13 (t, 2H), 4.55 (s, 2H), 6.07 (s, 1H), 6.65 (d, 1H), 6.77-6.83 (m, 4H), 7.01 (d, 1H), 7.06-7.14 (m, 3H), 7.27-7.38 (m, 3H), 7.54 (d, 1H), 7.68 (d, 1H), 7.73 (d, 1H), 7.89 (d, 1H), 8.15 (d, 1H)。
TLC:Rf 0.61(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.63 (s, 3H), 4.24-4.32 (m, 4H), 4.63 (s, 2H), 5.89-5.90 (m, 1H), 6.14 (s, 1H), 6.73 (d, 1H), 6.82-6.86 (m, 1H), 6.93-6.97 (m, 1H), 7.07 (d, 1H), 7.13 (d, 1H), 7.21 (d, 1H), 7.23-7.27 (m, 1H), 7.51-7.60 (m, 4H), 7.89-7.94 (m, 2H), 8.01-8.03 (m, 2H)。
TLC:Rf 0.67(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.35-1.41 (m, 8H), 2.44-2.50 (m, 4H), 2.63-2.66 (m, 5H), 3.92 (t, 2H), 4.69 (s, 2H), 6.49 (s, 1H), 6.72 (d, 1H), 6.81-6.84 (m, 1H), 6.94-6.98 (m, 1H), 7.13 (d, 1H), 7.21-7.25 (m, 1H), 7.51-7.59 (m, 4H), 7.87-7.92 (m, 2H), 8.03 (d, 1H), 8.11 (d, 1H)。
TLC:Rf 0.66(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.48 (s, 3H), 2.65 (s, 3H), 3.42 (t, 2H), 4.04 (t, 2H), 4.69 (s, 2H), 6.37 (s, 1H), 6.45 (d, 2H), 6.53-6.57 (m, 1H), 6.73 (d, 1H), 6.79-6.82 (m, 1H), 6.92-6.96 (m, 1H), 7.00-7.07 (m, 3H), 7.21-7.25 (m, 1H), 7.49-7.59 (m, 4H), 7.83 (d, 1H), 7.89 (d, 1H), 8.03-8.07 (m, 2H)。
TLC:Rf 0.52(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.63 (s, 3H), 4.14 (t, 2H), 4.24 (t, 2H), 4.66 (s, 2H), 6.31 (s, 1H), 6.65 (s, 1H), 6.73-6.75 (m, 2H), 6.84-6.87 (m, 1H), 6.93-6.97 (m, 1H), 7.11 (d, 1H), 7.16-7.18 (m, 1H), 7.23-7.27 (m, 1H), 7.53-7.59 (m, 4H), 7.90-7.94 (m, 2H), 8.01-8.06 (m, 2H)。
TLC:Rf 0.60(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.61 (s, 3H), 2.84 (t, 2H), 4.11 (t, 2H), 4.72 (s, 2H), 6.54 (s, 1H), 6.66 (d, 1H), 6.72-6.76 (m, 1H), 6.85-6.89 (m, 1H), 6.97 (d, 2H), 7.06 (d, 1H), 7.12-7.16 (m, 1H), 7.34-7.37 (m, 3H), 7.53-7.57 (m, 2H), 7.72-7.74 (m, 1H), 7.97 (d, 1H), 8.22-8.24 (m, 3H)。
TLC:Rf 0.63(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.64 (s, 3H), 3.09 (s, 3H), 3.21 (t, 2H), 3.30 (t, 2H), 3.52 (t, 2H), 4.00 (t, 2H), 4.70 (s, 2H), 6.42 (s, 1H), 6.72 (d, 1H), 6.81-6.84 (m, 1H), 6.93-6.97 (m, 1H), 7.15 (d, 1H), 7.21-7.25 (m, 1H), 7.50-7.59 (m, 4H), 7.85-7.91 (m, 2H), 8.04 (d, 1H), 8.10 (d, 1H)。
TLC:Rf 0.67(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.64 (s, 3H), 2.85 (t, 2H), 4.10 (t, 2H), 4.68 (s, 2H), 6.37 (s, 1H), 6.72 (d, 1H), 6.76 (d, 1H), 6.81-6.84 (m, 1H), 6.90-6.96 (m, 2H), 7.13 (d, 1H), 7.21-7.25 (m, 2H), 7.49-7.59 (m, 4H), 7.86 (d, 1H), 7.88 (d, 1H), 8.04-8.07 (m, 2H)。
TLC:Rf 0.68(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.64 (s, 3H), 3.05 (t, 2H), 4.05 (t, 2H), 4.67 (s, 2H), 6.38 (s, 1H), 6.61-6.62 (m, 1H), 6.72-6.76 (m, 2H), 6.81-6.84 (m, 1H), 6.93-6.97 (m, 1H), 7.11-7.14 (m, 2H), 7.21-7.25 (m, 1H), 7.50-7.59 (m, 4H), 7.86 (d, 1H), 7.90 (d, 1H), 8.03-8.06 (m, 2H)。
TLC:Rf 0.64(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.53 (s, 3H), 2.22 (t, 2H), 2.65 (s, 3H), 3.97 (t, 2H), 4.45 (s, 1H), 4.55 (s, 1H), 4.69 (s, 2H), 6.40 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.95-6.98 (m, 1H), 7.13 (d, 1H), 7.22-7.26 (m, 1H), 7.51-7.59 (m, 4H), 7.89 (d, 1H), 7.91 (d, 1H), 8.03 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.57(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.65 (s, 3H), 2.88-2.90 (m, 4H), 3.20 (t, 2H), 3.99 (t, 2H), 4.70 (s, 2H), 5.81 (s, 1H), 6.43 (s, 1H), 6.74 (d, 1H), 6.82-6.85 (m, 1H), 6.96-7.00 (m, 1H), 7.18 (d, 1H), 7.22-7.26 (m, 1H), 7.51-7.61 (m, 4H), 7.89-7.93 (m, 2H), 8.03 (d, 1H), 8.10 (d, 1H)。
TLC:Rf 0.63(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.23-2.29 (m, 2H), 2.64 (s, 3H), 3.92 (t, 2H), 4.69 (s, 2H), 4.77-4.85 (m, 2H), 5.50-5.61 (m, 1H), 6.40 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.94-6.98 (m, 1H), 7.13 (d, 1H), 7.22-7.26 (m, 1H), 7.51-7.59 (m, 4H), 7.87-7.92 (m, 2H), 8.03 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.67(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.29 (d, 3H), 2.23-2.28 (m, 2H), 2.65 (s, 3H), 3.89 (t, 2H), 4.70 (s, 2H), 5.12-5.17 (m, 1H), 5.23-5.28 (m, 1H), 6.38 (s, 1H), 6.72 (d, 1H), 6.81-6.85 (m, 1H), 6.94-6.98 (m, 1H), 7.14 (d, 1H), 7.22-7.26 (m, 1H), 7.52-7.60 (m, 4H), 7.88-7.92 (m, 2H), 8.03 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.60(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.91-1.98 (m, 2H), 2.50 (t, 2H), 2.66 (s, 3H), 3.92 (t, 2H), 4.71 (s, 2H), 6.42 (s, 1H), 6.73 (d, 1H), 6.80-6.83 (m, 1H), 6.95-6.97 (m, 2H), 7.08-7.11 (m, 2H), 7.20-7.24 (m, 1H), 7.49-7.60 (m, 5H), 7.86-7.90 (m, 2H), 8.02 (d, 1H), 8.12 (d, 1H), 8.37 (d, 1H)。
TLC:Rf 0.49(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.46-1.55 (m, 1H), 1.67-1.76 (m, 1H), 2.63 (s, 3H), 3.10-3.33 (m, 3H), 3.92-3.99 (m, 2H), 4.69 (s, 2H), 6.43 (s, 1H), 6.71 (d, 1H), 6.80-6.83 (m, 1H), 6.93-6.97 (m, 1H), 7.14 (d, 1H), 7.20-7.23 (m, 1H), 7.49-7.59 (m, 4H), 7.85 (d, 1H), 7.89 (d, 1H), 8.04-8.08 (m, 2H)。
TLC:Rf 0.43(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 1.00 (t, 6H), 2.68-3.00 (m, 9H), 4.08-4.10 (m, 2H), 4.51 (s, 2H), 6.61 (s, 1H), 6.69-6.72 (m, 1H), 6.79-6.81 (m, 1H), 7.04-7.05 (m, 2H), 7.13-7.18 (m, 1H), 7.38-7.50 (m, 3H), 7.68 (d, 1H), 7.76-7.80 (m, 2H), 7.85 (d, 1H), 8.19 (d, 1H)。
TLC:Rf 0.50(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.35-1.46 (m, 2H), 1.56-1.63 (m, 2H), 2.64 (s, 3H), 3.89 (t, 2H), 4.15-4.18 (m, 1H), 4.27-4.30 (m, 1H), 4.72 (s, 2H), 6.42 (s, 1H), 6.72 (d, 1H), 6.79-6.82 (m, 1H), 6.93-6.97 (m, 1H), 7.13 (d, 1H), 7.19-7.23 (m, 1H), 7.48-7.58 (m, 4H), 7.82 (d, 1H), 7.88 (d, 1H), 8.06 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.47(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.71-1.78 (m, 2H), 1.90-2.02 (m, 2H), 2.65 (s, 3H), 3.96 (t, 2H), 4.71 (s, 2H), 6.41 (s, 1H), 6.74 (d, 1H), 6.79-6.82 (m, 1H), 6.95-6.99 (m, 1H), 7.15 (d, 1H), 7.20-7.24 (m, 1H), 7.49-7.59 (m, 4H), 7.84 (d, 1H), 7.89 (d, 1H), 8.04 (d, 1H), 8.10 (d, 1H)。
TLC:Rf 0.24(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.18-1.27 (m, 2H), 1.52-1.60 (m, 2H), 2.63 (s, 3H), 3.27 (t, 2H), 3.86 (t, 2H), 4.71 (s, 2H), 6.42 (s, 1H), 6.70 (d, 1H), 6.77-6.81 (m, 1H), 6.91-6.96 (m, 1H), 7.11 (d, 1H), 7.17-7.20 (m, 1H), 7.45-7.52 (m, 3H), 7.56 (d, 1H), 7.77 (d, 1H), 7.84 (d, 1H), 8.05-8.10 (m, 2H)。
TLC:Rf 0.58(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.81-1.93 (m, 2H), 2.64 (s, 3H), 3.96 (t, 2H), 4.10-4.13 (m, 1H), 4.22-4.25 (m, 1H), 4.72 (s, 2H), 6.42 (s, 1H), 6.72 (d, 1H), 6.79-6.82 (m, 1H), 6.93-6.97 (m, 1H), 7.11 (d, 1H), 7.18-7.22 (m, 1H), 7.46-7.53 (m, 3H), 7.57 (d, 1H), 7.77 (d, 1H), 7.85 (d, 1H), 8.07-8.10 (m, 2H)。
TLC:Rf 0.61(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.26-1.33 (m, 6H), 2.64 (s, 3H), 3.09-3.16 (m, 1H), 3.34-3.38 (m, 1H), 3.65-3.90 (m, 3H), 4.72 (s, 2H), 6.37 (s, 1H), 6.71 (d, 1H), 6.80-6.83 (m, 1H), 6.92-6.96 (m, 1H), 7.14 (d, 1H), 7.21-7.25 (m, 1H), 7.50-7.60 (m, 4H), 7.86-7.92 (m, 2H), 8.04 (d, 1H), 8.09 (d, 1H)。
TLC:Rf 0.62(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.80-1.88 (m, 2H), 2.36 (t, 2H), 2.65 (s, 3H), 3.90 (t, 2H), 4.71 (s, 2H), 6.42 (s, 1H), 6.73 (d, 1H), 6.80-6.84 (m, 1H), 6.94-6.98 (m, 1H), 7.09 (d, 1H), 7.14-7.17 (m, 1H), 7.21-7.25 (m, 1H), 7.33 (d, 1H), 7.49-7.60 (m, 4H), 7.86-7.90 (m, 2H), 8.02 (d, 1H), 8.14 (d, 1H), 8.21 (s, 1H), 8.32 (d, 1H)。
TLC:Rf 0.62(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.81-1.88 (m, 2H), 2.34 (t, 2H), 2.66 (s, 3H), 3.89 (t, 2H), 4.72 (s, 2H), 6.41 (s, 1H), 6.73 (d, 1H), 6.81-6.84 (m, 1H), 6.93-6.98 (m, 3H), 7.10 (d, 1H), 7.22-7.25 (m, 1H), 7.49-7.61 (m, 4H), 7.86-7.90 (m, 2H), 8.02 (d, 1H), 8.13 (d, 1H), 8.32 (d, 2H)。
TLC:Rf 0.52(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 1.95-2.02 (m, 2H), 2.65 (s, 3H), 3.74 (t, 2H), 3.86 (t, 2H), 4.71 (s, 2H), 6.39 (s, 1H), 6.74 (d, 1H), 6.81-6.85 (m, 2H), 6.92 (s, 1H), 6.95-6.99 (m, 1H), 7.06 (d, 1H), 7.22-7.26 (m, 1H), 7.43 (s, 1H), 7.50-7.60 (m, 4H), 7.87-7.91 (m, 2H), 8.02 (d, 1H), 8.14 (d, 1H)。
グリシドールの代わりに、相当するエポキシド体を用いて、実施例30→実施例7と同様の操作を行って、以下の物性値を有する本発明化合物を得た。
TLC:Rf 0.49(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 0.86 (d, 3H), 2.63 (s, 3H), 3.73-3.81 (m, 3H), 4.70 (s, 2H), 6.47 (s, 1H), 6.68 (d, 1H), 6.76-6.79 (m, 1H), 6.90-6.93 (m, 1H), 7.13 (d, 1H), 7.16-7.20 (m, 1H), 7.43-7.50 (m, 3H), 7.55 (d, 1H), 7.73 (d, 1H), 7.82 (d, 1H), 8.06 (d, 1H), 8.11 (d, 1H)。
TLC:Rf 0.51(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 2.64 (s, 3H), 3.06-3.09 (m, 5H), 3.72-3.82 (m, 2H), 3.91-3.96 (m, 1H), 4.69 (s, 2H), 6.43 (s, 1H), 6.70 (d, 1H), 6.78-6.81 (m, 1H), 6.92-6.95 (m, 1H), 7.13 (d, 1H), 7.18-7.22 (m, 1H), 7.45-7.58 (m, 4H), 7.78 (d, 1H), 7.85 (d, 1H), 8.06-8.10 (m, 2H)。
TLC:Rf 0.49(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(DMSO-D6):δ 0.93 (s, 6H), 2.64 (s, 3H), 3.78 (s, 2H), 4.70 (s, 2H), 6.43 (s, 1H), 6.70 (d, 1H), 6.80-6.83 (m, 1H), 6.92-6.95 (m, 1H), 7.21-7.25 (m, 2H), 7.51-7.61 (m, 4H), 7.88-7.93 (m, 2H), 8.02 (d, 1H), 8,11 (d, 1H)。
TLC:Rf 0.59(クロロホルム:メタノール:アンモニア水=79:20:1);
1H-NMR(CDCl3):δ 2.68 (s, 3H), 3.51-3.67 (m, 2H), 3.73-3.90 (m, 1H), 3.95-4.09 (m, 2H), 4.58 (s, 2H), 6.46 (s, 1H), 6.61-6.70 (m, 4H), 6.77-7.17 (m, 8H), 7.33-8.03 (m, 6H)。
薬理実験例1:ヒトキマーゼ酵素阻害活性の測定
Human Chymase Pure(以下、キマーゼと略す。East Coast Biologics社)は、-20℃の冷凍庫にて保管し、使用直前に蒸留水にて希釈して使用した。N-Succinyl-Ala-Ala-Pro-Phe-p-nitroanilide(以下、ペプチド基質と略す。シグマアルドリッチ社)は、ジメチルスルホキシド(以下、DMSOと略す。)にて50mmol/L溶液を調製後、-20℃の冷凍庫にて保管した。本発明化合物および比較化合物は、DMSOにて10mmol/L溶液とし、-20℃の冷凍庫にて保管した。測定当日に、50%DMSOにて目的終濃度の10倍濃度となるように段階希釈を行い、本発明化合物溶液および比較化合物溶液を調製した。96ウェルマイクロプレートに、本発明化合物溶液および比較化合物溶液10μLとアッセイバッファー(50mmol/L Tris-HCl、150mmol/L NaCl、50 units/mL heparin(pH7.6))で希釈したペプチド基質溶液(最終濃度2.5mmol/L)を80μL加え、37℃で10分間インキュベートした。これに対して、最終濃度0.1μg/Lとなるようにキマーゼを10μL加え、酵素反応を開始した。405nmにおける吸光度を30秒間隔で5分間モニターし、反応速度(ΔmO.D./分)を測定した。コントロールウェルおよびブランクウェルには、本発明化合物溶液および比較化合物溶液の替わりに50%DMSOを10μL添加した。また、ブランクウェルにはキマーゼの替わりに蒸留水を10μL添加した。コントロールウェルの反応速度からブランクウェルの反応速度を差し引いた値を100%とし、本発明化合物および比較化合物が含まれたウェルの反応速度を百分率値で算出した。酵素活性を50%および90%阻害する本発明化合物濃度および比較化合物濃度をそれぞれ、IC50値およびIC90値として表示した。
HepG2細胞を15,000細胞/ウェルの細胞密度でコラーゲンコートした96ウエルプレートに播種し、37℃、5%CO2-95%空気の培養器中で一晩培養した。その後、本発明化合物および比較化合物を24時間、細胞に暴露させた。本発明化合物および比較化合物はDMSOに溶解後、Minimum essential medium(MEM)にL-グルタミン、非必須アミノ酸および1%ウシ胎児血清を添加した培養液で100倍希釈した。本発明化合物および比較化合物の処置濃度は、0、6.25、12.5、25、50および100μmol/Lとした。24時間暴露させた後に細胞中のATP濃度をCelltiter Glo luminescent assay kit(Promega社)を用いて測定し、本発明化合物および比較化合物の細胞毒性作用を評価した。測定キットのアッセイバッファーは細胞を溶解させ、ルシフェリン-ルシフェラーゼ酵素活性を指標に細胞から放出されたATP濃度を測定した。発光はSpectraMax plate reader(Molecular device)にて測定した。本発明化合物および比較化合物の細胞毒性作用は、発光を10%および50%抑制する濃度(IC10およびIC50)で表した。すなわち、HepG2細胞に対して、10%の細胞に毒性を及ぼす濃度としてIC10値、50%の細胞に毒性を及ぼす濃度として、IC50値を算出した。
本発明化合物および比較化合物(10mmol/L DMSO溶液)5μLを50%アセトニトリル水溶液195μLで希釈し、250μmol/Lの本発明化合物溶液および比較化合物溶液を作成した。ウォーターバスであらかじめ37℃に加温した反応用容器にNADPH Co factor(NADPH Regenerating System SolutionA(BD Bioscience社)およびNADPH Regenerating System SolutionB(BD Bioscience社)を0.1mol/L pH7.4リン酸緩衝液で、各々10倍および50倍希釈したもの(NADP+2.6mmol/L))を含む0.5mg/mLヒト肝ミクロソーム(0.1mol/L pH7.4リン酸緩衝液)245μLを添加し、5分間プレインキュベーション後、本発明化合物溶液および比較化合物溶液5μLを添加し、反応を開始した(最終濃度5μmol/L)。開始後すぐに20μLを採取し、I.S.(内標準物質(ワーファリン))を含むアセトニトリル180μLに添加して反応を停止した。この20μLに除蛋白用フィルター付プレート上で50%アセトニトリル水溶液180μLを加え、撹拌後、吸引ろ過して、ろ液を標準サンプルとした。反応溶液を15分間インキュベーション後、肝ミクロソーム溶液20μLを採取し、I.S.(内標準物質(ワーファリン))を含むアセトニトリル180μLに添加して反応を停止した。この20μLに除蛋白用フィルター付プレート上で50%アセトニトリル水溶液180μLを加え、撹拌後、吸引ろ過して、ろ液を反応サンプルとした。残存率(%)は、試料溶液5μLをLC-MS/MS(Thermo Scientific社製 Discovery Max)に注入し、反応サンプルのピーク面積比(本発明化合物および比較化合物/I.S.)を標準サンプルのピーク面積比で除した値を100倍して算出した。
Claims (15)
- 一般式(I)
R1は、水素原子、メチル基、トリフルオロメチル基、ジフルオロメチル基、フッ素原子、または塩素原子を表し、
R2は、水素原子、1~6個のR11で置換されていてもよいC1~10のアルキル基、1~6個のR11で置換されていてもよいC2~10のアルケニル基、または1~6個のR11で置換されていてもよいC2~10のアルキニル基を表し、
R11は、
(i)ハロゲン原子、
(ii)OR8(基中、R8は
(1)水素原子、
(2)C1~3のアルキル基、
(3)C1~3のハロアルキル基、
(4)C5~6の炭素環基、
(5)1~2個の窒素原子、1個の酸素原子および/または酸化されていてもよい1個の硫黄原子を含有する5~6員のヘテロ環基、または
(6)水酸基、C1~3のアルコキシ基、またはNR9R10で置換されたC1~3のアルキル基(基中、R9およびR10はそれぞれ独立して、水素原子、C1~3のアルキル基、またはフェニル基を表す。)、
(iii)NR9R10(基中、R9およびR10はそれぞれ独立して、水素原子、C1~3のアルキル基、またはフェニル基を表す。)、
(iv)ハロゲン原子、オキソ基、またはメチル基から選ばれる少なくとも1種の基で置換されていてもよいC5~6の炭素環基、または
(v)ハロゲン原子、オキソ基、またはメチル基から選ばれる少なくとも1種の基で置換されていてもよい、1~4個の窒素原子、1~2個の酸素原子および/または酸化されていてもよい1~2個の硫黄原子を含有する3~10員のへテロ環基を表し、
R3は、水素原子、メチル基、またはフッ素原子を表し、
R4は、水素原子、C1~2のアルキル基、C1~2のハロアルキル基、またはハロゲン原子を表し、
R5は、水素原子、C1~3のアルキル基、C1~3のハロアルキル基、またはハロゲン原子を表し、
R6は、水素原子、C1~3のアルキル基、C1~3のハロアルキル基、またはハロゲン原子を表し、
R7は、水素原子、C1~4のアルキル基、アリル基、トリクロロエチル基、ベンジル基、フェナシル基、p-メトキシベンジル基、トリチル基、または2-クロロトリチル基を表し、
pは、1~3の整数を表し、
qは、1~4の整数を表し、
rは、1~6の整数を表す。
ただし、p、q、およびrが、それぞれ2以上の整数を表す場合、R4、R5、およびR6は、それぞれ独立して、同一でも異なっていてもよい。)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグ。 - R2が、水素原子、1~6個のR12で置換されていてもよいC1~10のアルキル基、1~6個のR12で置換されていてもよいC2~10のアルケニル基、または1~6個のR12で置換されていてもよいC2~10のアルキニル基を表し、
R12が、ハロゲン原子、水酸基、ジメチルアミノ基、ピロリジニル基、N-メチルピロリジニル基、ピぺリジニル基、ハロゲン原子で置換されていてもよいフェノキシ基、ハロゲン原子で置換されていてもよいフェニル基、テトラヒドロピラニル基、2,3-ジヒドロベンゾフラニル基、チオフェニル基、ジイソプロピルアミノ基、メチル(フェニル)アミノ基、ピぺラジニル基、2-オキソ-1-ピロリジニル基、モルホリニル基、1,1-ジオキソチオモルホリニル基、イミダゾリル基、ピリジル基、2-メトキシエチレンオキシ基、ジメチルアミノエチレンオキシ基、ジエチルアミノエチレンオキシ基、1,2,3,4-テトラヒドロイソキノリニル基、アゼパニル基、ピラゾリル基、2-オキソイミダゾリジニル基、ジエチルアミノ基、メトキシ基、またはN-メチルピラゾリル基で示される請求項3記載の化合物。 - R2が、1~6個のR12で置換されているC1~6のアルキル基、1~6個のR12で置換されていてもよいC2~6のアルケニル基、または1~6個のR12で置換されていてもよいC2~6のアルキニル基を表し、R12が請求項4記載のR12と同じ意味である請求項3記載の化合物。
- R7が、水素原子で示される請求項4記載の化合物。
- 一般式(I)で示される化合物が、
(1) 2-{3-[(4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(2) 2-{3-[(1,4-ジメチル-1H-インドール-3-イル)メチル]-6-メチルイミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(3) 2-{3-[(1,4-ジメチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(4) 2-{3-[(1-エチル-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(5) 2-{6-フルオロ-3-[(4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(6) 2-{3-[(5-フルオロ-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}安息香酸、
(7) 2-[3-({4-メチル-1-[2-(4-モルホリニル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(8) 2-(3-{[4-メチル-1-(2-プロパニル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(9) 2-{3-[(4-メチル-1-プロピル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(10) 2-{3-[(1-エチル-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(11) 2-{3-[(5-フルオロ-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(12) 2-(3-{[1-(2-ヒドロキシエチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(13) 2-[3-({1-[3-(ジメチルアミノ)-2-ヒドロキシプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(14) 2-(3-{[1-(2,3-ジヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(15) 2-[3-({1-[2-(ジメチルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(16) 2-[3-({1-[3-(ジメチルアミノ)プロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(17) 2-[3-({4-メチル-1-[2-(ピロリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(18) 2-[3-({4-メチル-1-[2-(1-メチルピロリジン-2-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(19) 2-[3-({4-メチル-1-[2-(ピぺリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(20) 2-(3-{[4-メチル-1-(2-フェノキシエチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(21) 2-[3-({1-[(3R)-3-ヒドロキシ-3-フェニルプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(22) 2-[3-({1-[(3S)-3-ヒドロキシ-3-フェニルプロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(23) 2-[3-({1-[2-(4-フルオロフェニル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(24) 2-[3-({4-メチル-1-[2-(テトラヒドロ-2H-ピラン-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(25) 2-[3-({1-[2-(2,3-ジヒドロ-1-ベンゾフラン-5-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(26) 2-(3-{[4-メチル-1-(3-フェニルプロピル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(27) 2-(3-{[4-メチル-1-(ピぺリジン-4-イルメチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(28) 2-(3-{[1-(3-ヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(29) 2-[3-({1-[2-(ジプロパン-2-イルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(30) 2-[3-({4-メチル-1-[2-(ピペラジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(31) 2-[3-({4-メチル-1-[2-(2-オキソピロリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(32) 2-[3-({1-[2-(1,1-ジオキシドチオモルホリン-4-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(33) 2-[3-({1-[2-(3,4-ジヒドロイソキノリン-2(1H)-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(34) 2-{3-[(1-{2-[2-(ジエチルアミノ)エトキシ]エチル}-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(35) 2-[3-({4-メチル-1-[2-(1-メチル-1H-ピラゾール-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(36) 2-{3-[(1-{2-[2-(ジメチルアミノ)エトキシ]エチル}-4-メチル-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(37) 2-(3-{[4-メチル-1-(4-メチルペンタ-3-エン-1-イル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(38) 2-[3-({1-[2-(3-フルオロフェニル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(39) 2-[3-({4-メチル-1-[2-(1H-ピラゾール-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(40) 2-[3-({1-[2-(アゼパン-1-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(41) 2-{3-[(4-メチル-1-{2-[メチル(フェニル)アミノ]エチル}-1H-インドール-3-イル)メチル]イミダゾ[1,2-a]ピリジン-2-イル}-1-ナフトエ酸、
(42) 2-[3-({1-[2-(1H-イミダゾール-1-イル)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(43) 2-[3-({4-メチル-1-[2-(ピリジン-4-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(44) 2-[3-({1-[2-(2-メトキシエトキシ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(45) 2-[3-({4-メチル-1-[2-(チオフェン-3-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(46) 2-[3-({4-メチル-1-[2-(チオフェン-2-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(47) 2-(3-{[4-メチル-1-(3-メチルブタ-3-エン-1-イル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(48) 2-[3-({4-メチル-1-[2-(2-オキソイミダゾリジン-1-イル)エチル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(49) 2-(3-{[1-(ブタ-3-エン-1-イル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(50) 2-[3-({4-メチル-1-[(3Z)-ペンタ-3-エン-1-イル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(51) 2-[3-({4-メチル-1-[3-(ピリジン-2-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(52) 2-(3-{[1-(3,4-ジヒドロキシブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(53) 2-[3-({1-[2-(ジエチルアミノ)エチル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(54) 2-(3-{[1-(4-フルオロブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(55) 2-(3-{[4-メチル-1-(4,4,4-トリフルオロブチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(56) 2-(3-{[1-(4-ヒドロキシブチル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(57) 2-(3-{[1-(3-フルオロプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(58) 2-(3-{[4-メチル-1-(テトラヒドロ-2H-ピラン-2-イルメチル)-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(59) 2-[3-({4-メチル-1-[3-(ピリジン-3-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(60) 2-[3-({4-メチル-1-[3-(ピリジン-4-イル)プロピル]-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(61) 2-[3-({1-[3-(1H-イミダゾール-1-イル)プロピル]-4-メチル-1H-インドール-3-イル}メチル)イミダゾ[1,2-a]ピリジン-2-イル]-1-ナフトエ酸、
(62) 2-(3-{[1-(2-ヒドロキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(63) 2-(3-{[1-(2-ヒドロキシ-3-メトキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、
(64) 2-(3-{[1-(2-ヒドロキシ-2-メチルプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸、または
(65) 2-(3-{[1-(2-ヒドロキシ-3-フェノキシプロピル)-4-メチル-1H-インドール-3-イル]メチル}イミダゾ[1,2-a]ピリジン-2-イル)-1-ナフトエ酸である請求項6記載の化合物。 - 請求項1記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグを有効成分として含有してなる医薬組成物。
- キマーゼ阻害剤である請求項8記載の医薬組成物。
- キマーゼ介在性疾患の予防および/または治療剤である請求項8記載の医薬組成物。
- キマーゼ介在性疾患がアトピー性皮膚炎、潰瘍性大腸炎、心不全、肺線維症、動脈瘤、非アルコール性脂肪性肝炎、消化性潰瘍、またはアレルギー性結膜炎である請求項10記載の医薬組成物。
- 請求項1記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグと、非ステロイド系抗炎症薬、ステロイド薬、免疫抑制薬、プロスタグランジン類、抗アレルギー薬、メディエーター遊離抑制薬、ロイコトリエン受容体拮抗薬、抗ヒスタミン薬、オピオイド作用薬、ホスホジエステラーゼ阻害薬、フォルスコリン製剤、一酸化窒素合成酵素阻害薬、カンナビノイド-2受容体刺激薬、デコイ製剤、アミノサリチル酸製剤、利尿薬、アンジオテンシン変換酵素阻害薬、アンジオテンシンII受容体拮抗薬、抗不整脈薬、ジギタリス製剤、キマーゼ阻害薬、レニン阻害薬、カルシウム拮抗薬、ベータ遮断薬、硝酸薬、抗アルドステロン薬、強心薬、抗血小板薬、抗凝固薬、抗線維化薬、血糖降下薬、血圧低下薬、脂質改善薬、抗肥満薬、肝庇護薬、および抗酸化薬から選ばれる少なくとも1種以上と組み合わせてなる医薬。
- 請求項1記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグの有効量を患者に投与することを特徴とするキマーゼ介在性疾患の予防および/または治療方法。
- キマーゼ介在性疾患を予防および/または治療するための、請求項1記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグ。
- キマーゼ介在性疾患の予防および/または治療剤の製造のための請求項1記載の一般式(I)で示される化合物、その塩、そのN-オキシド体もしくはそれらの溶媒和物、またはそれらのプロドラッグの使用。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES10789507.0T ES2511065T3 (es) | 2009-06-17 | 2010-06-16 | Nuevo compuesto imidazopiridina |
| EP10789507.0A EP2444401B1 (en) | 2009-06-17 | 2010-06-16 | Novel imidazopyridine compound |
| JP2011519804A JP5621771B2 (ja) | 2009-06-17 | 2010-06-16 | 新規なイミダゾピリジン化合物 |
| US13/378,550 US8569504B2 (en) | 2009-06-17 | 2010-06-16 | Imidazopyridine compound |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009-143840 | 2009-06-17 | ||
| JP2009143840 | 2009-06-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010147133A1 true WO2010147133A1 (ja) | 2010-12-23 |
Family
ID=43356449
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/060172 WO2010147133A1 (ja) | 2009-06-17 | 2010-06-16 | 新規なイミダゾピリジン化合物 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US8569504B2 (ja) |
| EP (1) | EP2444401B1 (ja) |
| JP (1) | JP5621771B2 (ja) |
| ES (1) | ES2511065T3 (ja) |
| WO (1) | WO2010147133A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110483504A (zh) * | 2019-08-02 | 2019-11-22 | 桂林理工大学 | 以DMF为甲酰化试剂构建2-(2-萘基)咪唑[1,2-a]吡啶-3-醛的新方法 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2894174A1 (en) * | 2012-12-06 | 2014-06-12 | Regado Biosciences Inc. | Oligonucleotide formulation |
| CN110483506B (zh) * | 2019-08-02 | 2022-04-19 | 桂林理工大学 | 以DMF为甲酰化试剂构建2-(2-噻吩基)咪唑[1,2-a]吡啶-3-醛的新方法 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2868691A (en) | 1956-03-21 | 1959-01-13 | Riker Laboratories Inc | Self-propelling compositions for inhalation therapy containing a salt of isoproterenol or epinephrine |
| US3095355A (en) | 1961-10-12 | 1963-06-25 | Revlon | Aerosol composition |
| JPS52116494A (en) * | 1976-01-28 | 1977-09-29 | Gallardo Antonio Sa | Imidazo*1*22a*pyridine derivatives |
| JPH06184149A (ja) * | 1992-12-18 | 1994-07-05 | Asahi Glass Co Ltd | 新規イミダゾ[1,2−a]ピリジン誘導体 |
| JP2001199983A (ja) | 2000-01-18 | 2001-07-24 | Teijin Ltd | ベンズイミダゾール誘導体 |
| WO2001053272A1 (fr) * | 2000-01-17 | 2001-07-26 | Teijin Limited | Inhibiteurs de chymase humaine |
| WO2002010135A1 (fr) | 2000-08-01 | 2002-02-07 | Ono Pharmaceutical Co., Ltd. | Composes derives de 3,4-dihydroisoquinoline et medicaments contenant ces composes comme principe actif |
| WO2003064389A1 (fr) | 2002-01-31 | 2003-08-07 | Ono Pharmaceutical Co., Ltd. | Composes bicycliques contenant de l'azote et medicaments contenant ces composes en tant qu'ingredient actif |
| WO2008045688A1 (en) | 2006-10-06 | 2008-04-17 | Boehringer Ingelheim International Gmbh | Chymase inhibitors |
| WO2008153129A1 (ja) | 2007-06-14 | 2008-12-18 | Teijin Pharma Limited | 尿酸値低下剤 |
-
2010
- 2010-06-16 US US13/378,550 patent/US8569504B2/en not_active Expired - Fee Related
- 2010-06-16 EP EP10789507.0A patent/EP2444401B1/en not_active Not-in-force
- 2010-06-16 JP JP2011519804A patent/JP5621771B2/ja not_active Expired - Fee Related
- 2010-06-16 WO PCT/JP2010/060172 patent/WO2010147133A1/ja active Application Filing
- 2010-06-16 ES ES10789507.0T patent/ES2511065T3/es active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2868691A (en) | 1956-03-21 | 1959-01-13 | Riker Laboratories Inc | Self-propelling compositions for inhalation therapy containing a salt of isoproterenol or epinephrine |
| US3095355A (en) | 1961-10-12 | 1963-06-25 | Revlon | Aerosol composition |
| JPS52116494A (en) * | 1976-01-28 | 1977-09-29 | Gallardo Antonio Sa | Imidazo*1*22a*pyridine derivatives |
| JPH06184149A (ja) * | 1992-12-18 | 1994-07-05 | Asahi Glass Co Ltd | 新規イミダゾ[1,2−a]ピリジン誘導体 |
| WO2001053272A1 (fr) * | 2000-01-17 | 2001-07-26 | Teijin Limited | Inhibiteurs de chymase humaine |
| JP2001199983A (ja) | 2000-01-18 | 2001-07-24 | Teijin Ltd | ベンズイミダゾール誘導体 |
| WO2002010135A1 (fr) | 2000-08-01 | 2002-02-07 | Ono Pharmaceutical Co., Ltd. | Composes derives de 3,4-dihydroisoquinoline et medicaments contenant ces composes comme principe actif |
| WO2003064389A1 (fr) | 2002-01-31 | 2003-08-07 | Ono Pharmaceutical Co., Ltd. | Composes bicycliques contenant de l'azote et medicaments contenant ces composes en tant qu'ingredient actif |
| WO2008045688A1 (en) | 2006-10-06 | 2008-04-17 | Boehringer Ingelheim International Gmbh | Chymase inhibitors |
| WO2008153129A1 (ja) | 2007-06-14 | 2008-12-18 | Teijin Pharma Limited | 尿酸値低下剤 |
Non-Patent Citations (11)
| Title |
|---|
| "Comprehensive Organic Transformations : A Guide to Functional Group Preparations", 1999, JOHN WILEY & SONS INC |
| CARDIOVASCULAR RESEARCH, vol. 60, 2003, pages 413 - 420 |
| CIRCULATION RESEARCH, vol. 66, 1990, pages 883 |
| EUR. J. PHARMACOL., vol. 478, 2003, pages 179 - 185 |
| EUR. J. PHARMACOL., vol. 601, 2008, pages 186 - 191 |
| J. PHARMACOL. EXP. THER., vol. 324, 2008, pages 422 - 426 |
| RICHARD C. LAROCK: "A Guide to Functional Group Preparations", 1999, JOHN WILEY & SONS INC |
| RICHARD C. LAROCK: "Comprehensive Organic Transformations : A Guide to Functional Group Preparations", 1999, JOHN WILEY & SONS INC |
| See also references of EP2444401A4 * |
| T. W. GREENE: "Protective Groups in Organic Synthesis", 1999, WILEY |
| T.W. GREENE: "Protective Groups in Organic Synthesis", 1999, WILEY |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110483504A (zh) * | 2019-08-02 | 2019-11-22 | 桂林理工大学 | 以DMF为甲酰化试剂构建2-(2-萘基)咪唑[1,2-a]吡啶-3-醛的新方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2444401B1 (en) | 2014-09-03 |
| EP2444401A1 (en) | 2012-04-25 |
| JPWO2010147133A1 (ja) | 2012-12-06 |
| JP5621771B2 (ja) | 2014-11-12 |
| ES2511065T3 (es) | 2014-10-22 |
| EP2444401A4 (en) | 2012-11-07 |
| US8569504B2 (en) | 2013-10-29 |
| US20120094944A1 (en) | 2012-04-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI382984B (zh) | 雜環化合物 | |
| JP6148706B2 (ja) | 1H−ピラゾロ[3,4−b]ピリジンおよびその治療的使用 | |
| JP6042944B2 (ja) | Wntシグナル経路のインダゾール阻害剤およびその治療的使用 | |
| TWI567068B (zh) | Trk抑制化合物 | |
| CN110172051B (zh) | IRE-1α抑制剂 | |
| PT2841437T (pt) | Derivados de imidazotiadiazol e imidazopirazina como inibidores de recetor 4 ativado por protease (par4) para o tratamento de agregação plaquetária | |
| KR20180119582A (ko) | 라이실 옥시다제의 인돌 및 아자인돌 할로알릴아민 유도체 억제제 및 이의 용도 | |
| JP2015178457A (ja) | ピラゾロピリジン誘導体、またはその薬理学的に許容される塩 | |
| AU2009298006A1 (en) | Pyrazolo pyridine derivatives as NADPH Oxidase inhibitors | |
| JP2012521428A (ja) | 疼痛治療用のp2x3受容体アンタゴニスト | |
| CA2828349C (en) | Heterocyclic compounds for the inhibition of pask | |
| CA3018316C (en) | Griseofulvin compound | |
| TW201014840A (en) | Azole compound | |
| JP2017537948A (ja) | Nadphオキシダーゼ阻害剤としてのアミドチアジアゾール誘導体 | |
| RU2672468C1 (ru) | Производные аза-адамантанов и их применение | |
| US9745282B2 (en) | Indoline compounds as aldosterone synthase inhibitors | |
| JP5621771B2 (ja) | 新規なイミダゾピリジン化合物 | |
| DK2781508T3 (en) | NITROGEN CONCENTRATED RING COMPOUNDS FOR USE AS CRTH2 ANTAGONISTS | |
| RU2425832C2 (ru) | Гетеросоединение | |
| RU2772212C2 (ru) | Двухзамещенные соединения пиразола для лечения заболеваний | |
| CN114671856A (zh) | 多取代的尿嘧啶衍生物及其用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10789507 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2011519804 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010789507 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13378550 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |